HIV-specific ADCC: preventive and therapeutic vaccine potential
Ivan StratovDepartment of Microbiology and Immunology
Grattan Street, Parkville
Vic. 3010, Australia
Tel: +61 3 8344 9938
Fax: +61 3 8344 3846
Email: stratovi@unimelb.edu.au
Microbiology Australia 35(2) 105-106 https://doi.org/10.1071/MA14034
Published: 15 May 2014
HIV vaccines based on neutralising antibody and CD8 cytotoxic T-cell immunity have failed to induce protection in phase III clinical trials, while live attenuated HIV vaccines (although effective in simian models) are considered too dangerous to use in humans due to reversion to virulence. New strategies and lateral thinking are required to develop a safe and effective vaccine against HIV. Antibody dependent cellular cytotoxicity (ADCC) represents one such modality that provides potential advantages over previous modalities and may also have a role in efforts to cure HIV infection.
As of December 2012, the WHO and UNAIDS estimated that 35 million people were infected with HIV and while anti-retroviral medication (ARV) has greatly improved the health of these individuals, only 9.7 million people are actually on ARV therapy1. This has implications for both the health of those infected and the continuing spread of the pandemic globally2. Scientific efforts to develop both an effective HIV vaccine and cure remain global health priorities.
To date, only the Thai RV144 vaccine trial has shown any efficacy in preventing HIV acquisition (31% reduction; P = 0.04)3. Vaccines based on inducing neutralising antibody to envelope (Vaxgen) or T-cell immune responses have not been efficacious to date (STEp trial and HVTN 505)4–6; it is not clear whether such strategies will ever result in sufficiently robust immune responses to be effective. Post hoc analysis of the RV144 trial indicated that IgG binding antibodies directed at the V1-V2 region of the HIV envelope were associated with protection7; however, such binding antibodies are subject to immune escape8, much like neutralising antibodies targeting envelope protein. Binding antibodies tend to invoke elements of the innate immune system (e.g. phagocytosis, NK-cells and complement) to afford protection from pathogens and can be particularly useful for intracellular pathogens such as viruses, while neutralising antibodies tend to bind to free pathogens blocking their entry. In the case of HIV, this restricts their efficacy to surface envelope protein. These observations and empiric knowledge of the high mutation rate (and hence immune escape potential) of envelope indicate that vaccines based on immune responses only against HIV envelope protein may never be optimally effective.
Immune responses targeting more conserved portions of the HIV genome (such as the polymerase proteins: reverse transcriptase, protease and integrase) are thus more attractive and have underpinned the success story behind antiretroviral ARV medication. Although mutational escape can still occur to single agents, using 3-drug combinations usually leads to full replication suppression and is the current practice in managing HIV infection. Vaccination to induce CD8 T-cell responses targeting multiple internal HIV proteins had the potential to replicate this phenomenon, however the STEP trial, despite targeting Gag, Pol and Nef proteins and inducing reasonably strong CD8 CTL responses to multiple proteins did not afford protection9. Although the breadth of CTL responses generated by the STEP vaccine regimen was not optimal, it may also reflect the time needed for CTL memory cells to be expanded within regional lymph nodes upon exposure to HIV-1, during which time mutational escape can be established. Furthermore, the effectiveness of CTL responses is highly HLA dependent10, thus limiting their protective efficacy in outbred human populations.
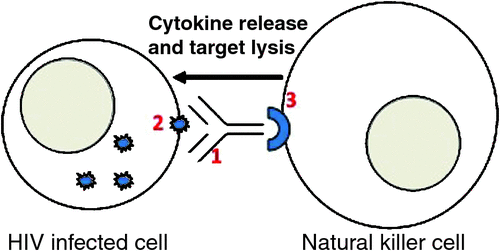
Antibody-dependent cellular cytotoxicity (ADCC) is an immune mechanism that allows antigen-specific targeting of HIV-infected cells by the innate immune system. Specifically, ADCC antibodies bind to antigens expressed on virus-infected cells and (via their Fc-receptor) recruit NK-cells inducing cytokine release by NK-cells and lysis of virus-infected cell (Figure 1).
|
In recent years our group has described a method of (a) identifying which HIV-infected subjects display HIV-specific ADCC, (b) quantifying the magnitude and cytokine profile of the response and (c) defining the exact antigenic target of the response11. We have shown that HIV-specific ADCC responses do target internal and regulatory HIV proteins such as Pol12 and Vpu13, in addition to targeting Envelope. Vpu-specific responses are, in fact, enriched in people with slowly progressing HIV infection13. We have shown that HIV-specific ADCC targeting envelope protein is stronger and broader in people with slowly progressing HIV (unpublished data), although such responses are (like neutralising antibody and CD8 T-cell responses) subject to immune escape14.
The ability of ADCC to target highly conserved internal HIV proteins (such as integrase12) is very attractive in a preventive vaccine strategy, although it remains unclear how these antigens are expressed by virus-infected cells. In terms of a therapeutic vaccine modality, HIV-specific ADCC has other potential advantages including the ability to target virus-infected cells (rather than free virions), the fact that ADCC is not restricted by HLA phenotype and ADCC can act at mucosal sites rather than in regional lymph nodes. We are proposing trialling passive vaccines strategies in macaques to evaluate the preventive efficacy of HIV-specific ADCC. HIV-specific ADCC was induced in the partially effective RV144 trial and correlated with reduced risk of infection7. The ability to induce effective ADCC antibodies by vaccination may be due to a reduced need for somatic mutation of immunoglobulin chains, compared to the hypermutation described for broadly effective neutralising antibodies15
We have also recently proposed a speculative but as yet untested additional role for HIV-specific ADCC, as part of efforts to cure HIV infection16. Researchers have sought ways to induce cells latently infected with HIV to express viral antigens and then use the immune system to clear the viral reservoir and effect a cure17. ADCC antibodies are potentially an attractive vehicle to promote clearance of latently infected cells, as an alternative to previously described (but ineffective) cytotoxic T-cell responses18.
In summary, ADCC antibodies to both Envelope and conserved internal proteins of HIV are attractive immune responses to induce by vaccination and may be able to improve upon the success of the Thai RV144 trial. Future research in my lab over the coming years will aim to isolate, purify and test the preventive and therapeutic potential of HIV-specific ADCC antibodies.
References
[1] WHO (2014) When you have hope. http://www.who.int/hiv/en/[2] Cohen, M.S. et al. (2011) Prevention of HIV-1 infection with early antiretroviral therapy. N. Engl. J. Med. 365, 493–505.
| Prevention of HIV-1 infection with early antiretroviral therapy.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3MXhtVars7jF&md5=f9b5762c300641977a5b0acaf07eb30fCAS | 21767103PubMed |
[3] Rerks-Ngarm, S. et al. (2009) Vaccination with ALVAC and AIDSVAX to prevent HIV-1 infection in Thailand. N. Engl. J. Med. 361, 2209–2220.
| Vaccination with ALVAC and AIDSVAX to prevent HIV-1 infection in Thailand.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD1MXhsFWhsb%2FO&md5=975d5fe43a25c7b782c45db3b6e74b5cCAS | 19843557PubMed |
[4] Buchbinder, S.P. et al. (2008) Efficacy assessment of a cell-mediated immunity HIV-1 vaccine (the Step Study): a double-blind, randomised, placebo-controlled, test-of-concept trial. Lancet 372, 1881–1893.
| Efficacy assessment of a cell-mediated immunity HIV-1 vaccine (the Step Study): a double-blind, randomised, placebo-controlled, test-of-concept trial.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD1cXhsVelurfM&md5=77a1d1972d9600669aca3efabe3e53e9CAS | 19012954PubMed |
[5] Gilbert, P.B. et al. (2005) Correlation between immunologic responses to a recombinant glycoprotein 120 vaccine and incidence of HIV-1 infection in a phase 3 HIV-1 preventive vaccine trial. J. Infect. Dis. 191, 666–677.
| Correlation between immunologic responses to a recombinant glycoprotein 120 vaccine and incidence of HIV-1 infection in a phase 3 HIV-1 preventive vaccine trial.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD2MXisVensbk%3D&md5=bcd49af781ec1100e2da7306c252ef8fCAS | 15688279PubMed |
[6] Hammer, S.M. et al. (2013) Efficacy trial of a DNA/rAd5 HIV-1 preventive vaccine. N. Engl. J. Med. 369, 2083–2092.
| Efficacy trial of a DNA/rAd5 HIV-1 preventive vaccine.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3sXhvFWqtr%2FF&md5=0e5837eb70df2d13806d937e3686688fCAS | 24099601PubMed |
[7] Haynes, B.F. et al. (2012) Immune-correlates analysis of an HIV-1 vaccine efficacy trial. N. Engl. J. Med. 366, 1275–1286.
| Immune-correlates analysis of an HIV-1 vaccine efficacy trial.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC38XlsVSmtL4%3D&md5=598d205801bf3ee99c1d7d68f2fbef4bCAS | 22475592PubMed |
[8] Roederer, M. et al. (2014) Immunological and virological mechanisms of vaccine-mediated protection against SIV and HIV. Nature 505, 502–508.
| Immunological and virological mechanisms of vaccine-mediated protection against SIV and HIV.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2cXhtFOgurk%3D&md5=fc9f6f3e04530cd9267162ecf708161fCAS | 24352234PubMed |
[9] McElrath, M.J. et al. (2008) HIV-1 vaccine-induced immunity in the test-of-concept step study: a case-cohort analysis. Lancet 372, 1894–1905.
| HIV-1 vaccine-induced immunity in the test-of-concept step study: a case-cohort analysis.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD1cXhsVelurfN&md5=f59f08f579e254e93273e21a5c2c6d70CAS | 19012957PubMed |
[10] Payne, R.P. et al. (2014) Differential escape patterns within the dominant HLA-B*57:03-restricted HIV Gag epitope reflect distinct clade-specific functional constraints. J. Virol. , .
| Differential escape patterns within the dominant HLA-B*57:03-restricted HIV Gag epitope reflect distinct clade-specific functional constraints.Crossref | GoogleScholarGoogle Scholar | 24501417PubMed |
[11] Stratov, I. et al. (2008) Robust NK cell-mediated human immunodeficiency virus (HIV)-specific antibody-dependent responses in HIV-infected subjects. J. Virol. 82, 5450–5459.
| Robust NK cell-mediated human immunodeficiency virus (HIV)-specific antibody-dependent responses in HIV-infected subjects.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD1cXmt1Sls7Y%3D&md5=58e696faa7d9ffff7c7518b790752c7eCAS | 18353957PubMed |
[12] Isitman, G. et al. (2011) Pol as a target for antibody dependent cellular cytotoxicity responses in HIV-1 infection. Virology 412, 110–116.
| Pol as a target for antibody dependent cellular cytotoxicity responses in HIV-1 infection.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3MXjtFertbg%3D&md5=64c90ba7b6542dd8ad62f26c9bdd35ceCAS | 21269655PubMed |
[13] Wren, L.H. et al. (2013) Specific antibody-dependent cellular cytotoxicity responses associated with slow progression of HIV infection. Immunology 138, 116–123.
| Specific antibody-dependent cellular cytotoxicity responses associated with slow progression of HIV infection.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3sXnslSrsw%3D%3D&md5=b0141de38e382245267b00dec5def7c2CAS | 23173935PubMed |
[14] Chung, A.W. et al. (2011) Immune escape from HIV-specific antibody-dependent cellular cytotoxicity (ADCC) pressure. Proc. Natl. Acad. Sci. USA 108, 7505–7510.
| Immune escape from HIV-specific antibody-dependent cellular cytotoxicity (ADCC) pressure.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3MXmtFSrsb0%3D&md5=5236d1767471ac4f48a1c98ea6aa2812CAS | 21502492PubMed |
[15] Bonsignori, M. et al. (2012) Antibody-dependent cellular cytotoxicity-mediating antibodies from an HIV-1 vaccine efficacy trial target multiple epitopes and preferentially use the VH1 gene family. J. Virol. 86, 11521–11532.
| Antibody-dependent cellular cytotoxicity-mediating antibodies from an HIV-1 vaccine efficacy trial target multiple epitopes and preferentially use the VH1 gene family.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC38XhsFCmur7E&md5=1f5b55e8f734aadf3096332f2781b1d1CAS | 22896626PubMed |
[16] Wren, L.H. et al. (2013) Obstacles to ideal anti-HIV antibody-dependent cellular cytotoxicity responses. Vaccine 31, 5506–5517.
| Obstacles to ideal anti-HIV antibody-dependent cellular cytotoxicity responses.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3sXhsVSns7bE&md5=ba3aa4587a6e89dab14930b30439ead7CAS | 23981432PubMed |
[17] Archin, N.M. and Margolis, D.M. (2014) Emerging strategies to deplete the HIV reservoir. Curr. Opin. Infect. Dis. 27, 29–35.
| Emerging strategies to deplete the HIV reservoir.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3sXitVSqtLnN&md5=a6b3202cbcbc7da919d50fdbe75e6dcdCAS | 24296585PubMed |
[18] Casazza, J.P. et al. (2013) Therapeutic vaccination expands and improves the function of the HIV-specific memory T-cell repertoire. J. Infect. Dis. 207, 1829–1840.
| Therapeutic vaccination expands and improves the function of the HIV-specific memory T-cell repertoire.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3sXotlOgur4%3D&md5=7c85931e21b5a0d63652a6cfd9d2e4e6CAS | 23482645PubMed |
Biography
Dr Stratov is a clinician-scientist, who has successfully taken a serendipitous observation in one patient through 10 years of rigorous scientific evaluation, across two cohorts of HIV-infected individuals, to establish his hypotheses regarding the biological importance of HIV-specific ADCC. His research plan has been methodical and resulted in 16 peer-reviewed scientific publications since 2008, culminating in a critical publication in the prestigious scientific journal Proceedings of the National Academies of Science, detailing immune escape from HIV-specific ADCC, and clinching the biological significance of HIV-specific ADCC. He has co-authored five reviews in the area of HIV-specific ADCC and received the 2013 ASID Frank Fenner award for his post-doctoral research.


