Battling habitat loss: suitability of anthropogenic waterbodies for amphibians associated with naturally acidic, oligotrophic environments
Clay Alan Simpkins A B D , James Guy Castley A , Jonathan D. Shuker
A B D , James Guy Castley A , Jonathan D. Shuker  A , Clare Morrison A and Jean-Marc Hero C
A , Clare Morrison A and Jean-Marc Hero C
A Environmental Futures Research Institute, Griffith University, Gold Coast campus, Parklands Drive, Qld 4222, Australia.
B Present address: Mackay North State High School, North Mackay, Qld 4740, Australia.
C School of Science and Engineering, the University of the Sunshine Coast, Maroochydore, Qld 4558, Australia.
D Corresponding author. Email: claysimpkins0@gmail.com
Pacific Conservation Biology 28(2) 174-183 https://doi.org/10.1071/PC20098
Submitted: 11 December 2020 Accepted: 24 May 2021 Published: 17 June 2021
Journal Compilation © CSIRO 2022 Open Access CC BY
Abstract
Habitat destruction is a key threatening process for amphibians. Modified or anthropogenic waterbodies can be used to compensate for habitat loss, with several amphibian species utilising created or modified waterbodies. We measured usage of anthropogenic/modified waterbodies by adult and tadpole frog species, including threatened species, in coastal wallum habitat in eastern Australia. Nine road trenches/ditches, 8 artificial ‘lakes’, 6 golf course waterbodies and 13 natural waterbodies were surveyed for frog adults and tadpoles during the summer/spring period 2011–2012. Additionally, we examined the relationship between frog assemblages and environmental factors (water chemistry, aquatic predators, vegetation types), focusing on Litoria olongburensis and Litoria fallax. Frog species richness differed between waterbody types, with two of three threatened frog species present in both natural and anthropogenic/modified waterbodies. The frog assemblage was influenced by pH, turbidity, salinity and percentage cover of certain vegetation types. Measured abundance of L. olongburensis adults and tadpoles were highest in natural waterbodies with high sedge density and low pH. The measured abundance of adult L. fallax was highest within artificial lakes. We conclude that differences in water chemistry and vegetation density can affect the amphibian assemblage within these environments. Therefore, effective conservation of all frog species would be enhanced by conserving a variety of waterbody types. However, natural waterbodies are more likely to be used by threatened species and the conservation of these waterbodies provides the best conservation outcomes. Where habitat offsets may be required, environmental managers should critically assess the quality of constructed habitat for specialist frog species including those associated with acidic, oligotrophic environments.
Keywords: anuran, frog, habitat modification, Litoria fallax, Litoria olongburensis, vegetation, water chemistry.
Introduction
Amphibian species are declining globally (Stuart et al. 2004), with habitat loss/modification being one of the main threatening factors (Collins and Storfer 2003; Beebee and Griffiths 2005; Nowakowski et al. 2017; Pyron 2018). This habitat loss could be compensated for by modified or anthropogenic waterbodies (e.g. Mazerolle et al. 2006; Ruhí et al. 2012), with several amphibian species within Australia (Hazell et al. 2004; Sievers et al. 2018), North America (Monello and Wright 1999; Brand and Snodgrass 2010; Brown et al. 2012) and Europe (Rannap et al. 2009; Brown et al. 2012; Ruhí et al. 2012; Magnus and Rannap 2019) utilising anthropogenic or modified waterbodies that were either specifically designed to compensate for habitat loss or were opportunistically used by amphibians. However, species assemblages and richness can differ between anthropogenic/modified and natural waterbodies (Hazell et al. 2004; Magnus and Rannap 2019), potentially due to differences in environmental factors that influence amphibian species distributions within waterbodies. Additionally, waterbodies that are used may be considered as ecological traps, and may not be suitable for long term amphibian populations. For the purpose of this study, an ecological trap is referred to as a habitat/waterbody that an individual frog occupies where it could perform poorly (e.g. survivability, successful reproduction) when compared with natural habitats (Robertson and Hutto 2006).
Several ecological interactions can influence the distribution of amphibians within waterbodies, including aquatic predators (Kats et al. 1988; Hero et al. 2001; Vonesh et al. 2009; Barry 2015), and/or competitors (Morin 1986; Wilbur 1987; Mokany and Shine 2003; Twomey et al. 2008). Water chemistry variables, including pH (reviewed in Pierce 1985; Sparling 2010) and salinity (Strahan 1957; Christy and Dickman 2002; Chinathamby et al. 2006; Rios-López 2008) may potentially lead to the exclusion of amphibian species from a waterbody due to tadpole water chemistry intolerances. Furthermore, turbidity may have an influence on structuring amphibian assemblages (Hecnar and M’Closkey 1996) while the abundance of particular vegetation species (Shuker and Hero 2013) or the proportion of the water margin with emergent vegetation (Hazell et al. 2004) can also have the ability to influence amphibian species richness or abundances within waterbodies.
Reductions in the availability of coastal waterbodies have occurred globally (e.g. Turner 1990; Levin et al. 2009), due to human population growth and expansion. Within Australia, the majority of freshwater, coastal waterbodies situated along the eastern seaboard between Fraser Island, Queensland, and Jervis Bay, New South Wales, are unique as they are both naturally oligotrophic (Hines et al. 1999) and acidic (Griffith et al. 2008; Hines and Meyer 2011; Simpkins et al. 2014; Shuker et al. 2016). The vegetation community within this area is known as ‘wallum’, and is defined as the vegetation communities that include Banksia woodland, sedgeland, heathland and Melaleuca swamps (Hines et al. 1999; Griffith et al. 2003). Populations of threatened ‘acid’ frog species (Ingram and Corben 1975) occur within wallum waterbodies (Hines et al. 1999; Lewis and Goldingay 2005) and areas where multiple acid frogs species may occur outside of protected areas have been identified (Filer et al. 2019). Furthermore, more than 30% of original heathland and Melaleuca cover in south-east Queensland was cleared between 1974 and 1989 (Catterall and Kingston 1993, cited in Hines et al. 1999) and areas within the acid frog species ranges have one of the highest human growth rates within Australia (Hines et al. 1999; Garden et al. 2010). Wallum habitats also contain modified and anthropogenic waterbodies (Simpkins 2012), which may be utilised by ‘acid’ frog species and constructed ‘compensatory’ waterbodies have been created to offset the loss of wallum ponds used by acid frogs. Therefore, these waterbodies could aid in reducing the impacts of habitat loss. Despite compensatory habitats being used to offset habitat and the presence of anthropogenic/modified waterbodies in wallum habitat, no peer-reviewed studies have examined whether frogs normally associated with naturally acidic, oligotrophic waterbodies use and successfully reproduce within these anthropogenic/modified waterbodies.
Consequently, we aimed to compare usage of natural and anthropogenic waterbodies by the adults and tadpoles of frog species within the coastal wallum of eastern Australia. This study allows us to (1) identify which environmental variables influence the frog assemblages within the various waterbodies of this unique habitat; and (2) determine the environmental variables that influence the relative abundance of the threatened wallum sedge frog, Litoria olongbureneis, and a potential competitor, Litoria fallax (Meyer et al. 2006), across the natural/modified landscape. The outcomes of this study can help environmental managers creating habitat offsets to critically assess the quality of constructed habitat for frog species, including threatened, specialist species, which are associated with naturally acidic, oligotrophic waterbodies, and their respective competitors. The results will also help determine whether human intervention through anthropogenic waterbody construction can compensate for habitat loss of specialist frogs that are unique to environments with low pH and nutrients.
Materials and methods
Study site selection and sampling design
Waterbodies were located within and around Tyagarah Nature Reserve (NR) (28.6067°S, 153.5693°E) and the southern section of Bribie Island National Park (NP) (27.0732°S, 153.1774°E). Waterbodies were selected using Google Earth satellite imagery to cover the style of waterbodies available at each site. All waterbodies were surveyed twice, first between 18 October 2011 and 5 December 2011, and again between 7 and 21 February 2012. Surveys for adult frogs, aquatic predators and tadpoles were conducted at 13 natural (n = 5 Tyagarah, n = 8 Bribie Island) and 25 anthropogenic waterbodies (total n = 38). Anthropogenic waterbodies consisted of roadside trenches/ditches (n = 4 Tyagarah; n = 5 Bribie Island), artificial ‘lakes’ (n = 1 Tyagarah; n = 8 Bribie Island) and golf course waterbodies (n = 6 Tyagarah). With the exception of golf course waterbodies, all waterbodies contained natural vegetation around the majority (>80%) of the waterbody perimeter.
Roadside trenches were classified as waterbodies that had been constructed next to roads or firebreaks where earth material had been removed to aid construction of the road or firebreak. Artificial lakes were classified as constructed waterbodies with the majority of the waterbody (>80%) as open water with vegetation fringing the perimeter. Golf course waterbodies were constructed waterbodies within the golf course boundaries and contained little vegetation around the perimeter (<10%). Waterbodies within wallum heathland can be composed of heterogeneous vegetation types. Therefore, one large natural waterbody had multiple transects established a minimum of 300 m apart to ensure transect independence. Survey transects were established 5 m from the waterbody perimeter and ran parallel to the waterbody perimeter. Transect length varied between 50 and 100 m, depending on waterbody size, with frogs being recorded 2 m on either side of the transect.
Litoria olongburensis appears to delay breeding to longer hydroperiods and can also respond quickly to wetland filling/inundation (Lowe et al. 2015). Hence, surveying was undertaken when waterbodies were within these ideal scenarios. Rainfall data was collected from the Australian Bureau of Meteorology Weather Station #058 216 for Tyagarah NR and #040 998 for Bribie Island NP. Sampling was conducted in 2011, with 246.9 mm rainfall recorded at Bribie Island and 373.4 mm recorded for Tyagarah NR during the 3 months prior to the date of the first survey. The 2012 surveys recorded 936.7 mm at Bribie Island and 405.2 mm for Tyagarah NR during the 3 months prior to the second survey date. All waterbodies surveyed had water present during both the 2011 and 2012 surveys.
Diurnal aquatic predator traps were placed at the beginning, middle and end of each transect. Traps were baited with Orca Floating Fish Food Pellets and were left for approximately 30 min before being collected. Diurnal dip-netting for predatory fauna and tadpoles was conducted using a circular net, with an opening of approximately 30 cm in diameter and mesh size <0.5 mm. Five ‘sweeps’ of the dip-net and measurements of pH and salinity were undertaken at 10 m sampling intervals along each transect. Each ‘sweep’ encompassed three water column levels (bottom, middle, and top) to capture any variation in tadpole species richness or abundance that may occur along the water depth gradient (Heyer 1973). Water columns were deep enough to be reached at all parts of the transect.
Water pH and salinity were measured using a TPS Aqua-CPA Conductivity-TDS-pH-Temperature Meter (ver. 1.2) approximately 15 cm below the surface of the water at every 10 m sampling point along the transect. Two 25 mL surface water samples were collected at the beginning and the end of each transect. Turbidity was measured from the 25 mL water samples using a HACH DREL 2000 Direct Reading Spectrophotometer within 24 h of sample collection. Water chemistry measurements and samples were taken before dip-netting and trapping were conducted. Waterbody area was calculated using Google Earth satellite imagery.
Vegetation on the transect was categorised into five vegetation types; sedge, grass, lily, Gahnia spp. and Melaleuca spp. Percent density of each vegetation type was visually estimated using a 1 × 1 m quadrat centered at each 10 m sampling interval.
Nocturnal frog surveys were conducted by walking and visually searching the transect using a Princeton Apec Headtorch. The species and the number of individuals seen for each species were recorded for each transect. For Crinia tinnula, numbers of individuals were determined using acoustic calls due to visual observations being absent or low at the survey transects. The vegetation type an individual frog was first encountered on was also recorded. Substrate records were only conducted on the first survey.
Data analysis
The number of L. olongburensis visually recorded during the first survey was tallied for each transect and divided by the transect length to obtain a relative abundance of L. olongburensis per metre. All other species recorded visually in sufficient numbers were counted and divided by the transect length.
Visual incidental and transect observations and frogs heard calling <1 m outside the waterbody perimeter or calling within the waterbody during the first or second survey were recorded as occupying the waterbody. Tadpole species were considered present within a waterbody if they were recorded from any of the sampling points.
Predatory fish caught in aquatic traps were used in data analysis due to low counts of predatory fish caught during dip-netting. The number of fish recorded for a given transect was divided by the number of aquatic traps used within the waterbody to obtain a relative fish abundance for each transect.
Salinity, turbidity and pH were averaged for each transect. Mean density of each vegetation type for each transect was calculated by dividing the total percentage density of each vegetation type from each sampling point by the number of sampling points along the transect.
A Spearman Rank Correlation Test was performed in IBM SPSS Statistics ver. 19 (SPSS Inc., Chicago, IL, USA) to determine correlated variables. Highly correlated variables were considered to be variables that had a correlation coefficient value ≥0.7 (sensu Garden et al. 2007; Simpkins et al. 2014). No variables were highly correlated and all variables were used in analyses.
A one-way ANOVA with Tukey’s post-hoc analysis was used to determine significant differences in environmental variables between waterbody types. One-way ANOVAs were also used to compare the relative abundance of L. olongburensis and L. fallax in the four different waterbody types.
The frog community assemblage was analysed (using visual counts for L. olongburensis, L. fallax, L. peronii, R. marina and L. tyleri, and acoustic counts for C. tinnula) using non-metric multidimensional scaling (nMDS). These species were chosen as they were encountered in at least two waterbodies and were highest in relative abundance when compared with other species. Only waterbodies which contained one or more of these six species were used in nMDS analysis (n = 27). Bray–Curtis distance measures were used to determine waterbody similarities for the relative abundance of individual species. Random permutations (n = 999) were used to determine which environmental variables were significantly influencing the amphibian assemblage (Oksanen 2011). Analyses were performed using the statistical program R (R Core Development Team 2011) using the vegan (ver. 2.04) package (Oksanen et al. 2012).
Twenty-eight models focusing on the influence of variables on the relative abundance of L. olongburensis and L. fallax were constructed a priori. To obtain the maximum number of predictor variables to use in each model, a generalised ‘rule of thumb’ of n/3 (where n = number of waterbodies sampled) (Crawley 2007) was applied. Predictor variables in L. olongburensis models were average pH, average salinity, average turbidity, waterbody size, fish relative abundance, L. fallax relative abundance, % sedge density, % Gahnia density, % grass density, % lily density, and % Melaleuca density. Predictor variables in L. fallax models were the same as the L. olongburensis models, except L. fallax relative abundance was replaced with L. olongburensis relative abundance. Generalised additive mixed effects models, using a Quasi-Poisson link function to account for overdispersion and location as a covariate, were used to determine the importance of the predictor variables on L. olongburensis and L. fallax relative abundance.
Small sample Akaike’s information criterion (AICc) was used for model selection, with the ‘best’ model having the lowest AICc value (Burnham and Anderson 2002). To determine the ranking of the models, ΔAICci values were calculated, where higher ΔAICc values indicated less accurate models for the given data (Burnham and Anderson 2002; Johnson and Omland 2004). If a model had a Δi ≤ 2, then there was considerable evidence that the model could be the ’best’ model, given the data (Johnson and Omland 2004). If a model had a Δi ≤ 2–4, then there was moderate evidence that the model could be the ‘best’ model, given the data. Each model was assigned a model weight (wi) which was used to determine the ‘probability that model I is the best model for the observed data, given the candidate set of models’ (Johnson and Omland 2004). The closer the wi was to 1, the closer the model was considered the best model for the given data (Burnham and Anderson 2002). To determine the relative importance of variables within models where Δi < 4, the wi values were summed from all models where Δi < 4 and where the variable of interest occurred. The closer the relative importance value was to 1, the higher the relative importance of the variable (Grueber et al. 2011). All models were run in the freeware statistical package R (R Core Development Team 2011) using the MuMIn (ver. 1.7.2) (Barton 2012) and vegan (ver. 2.04) (Oksanen et al. 2012) packages.
Results
Waterbody characteristics and predatory fish
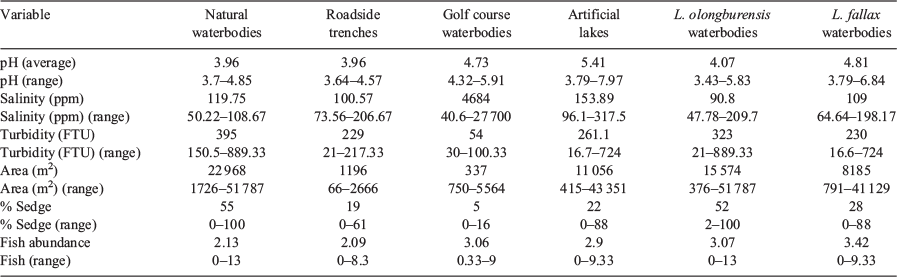
Waterbody characteristics varied among waterbody types (Table 1). Variables that were significantly different between waterbody types were pH (df = 36, F2/34= 5.36, P = 0.004), turbidity (df = 36, F2/34 = 3.925, P = 0.017) and waterbody size (df = 36, F2/34 = 6.001, P = 0.002). Tukey’s post-hoc analysis revealed pH was significantly higher in artificial lakes than in natural waterbodies (P = 0.005) and roadside ditches (P = 0.011). Tukey’s post-hoc analysis also revealed turbidity was significantly higher in natural waterbodies than in golf course waterbodies (P = 0.011) and natural waterbodies were significantly larger then road side ditches (P = 0.003) and golf course waterbodies (P = 0.024). The number of predatory fish in each waterbody included individuals from the species Gambusia holbrooki (Eastern mosquito fish), Rhadinocentrus ornatus (Ornate rainbow fish), Hypseleotris galii (Firetail gudgeon), Hypseleotris compressa (Empire gudgeon), and Hypseleotris sp. (Midgleys carp gudgeon).
|
Frog assemblage/occupancy
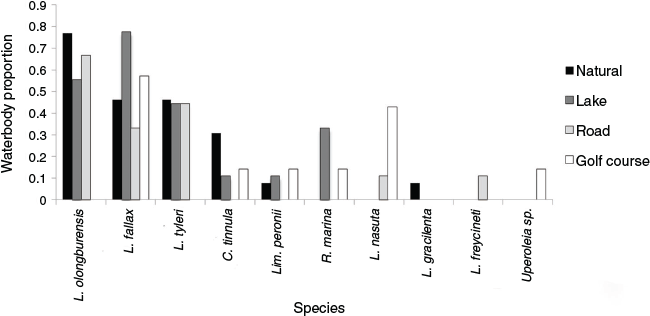
A total of 10 adult frog species were encountered; six recorded in natural waterbodies, six in artificial lakes, five in roadside ditches and six in golf course waterbodies (Fig. 1). Relative abundances of L. olongburensis and L. fallax adults were higher during the 2011 survey. Litoria olongburensis and C. tinnula occupancy was highest within natural waterbodies while L. fallax, L. tyleri and R. marina occupancy was highest within artificial lakes. Litoria freycineti was only recorded from roadside ditches while L. gracilenta was only recorded from natural waterbodies. Additionally, one Uperolia sp. was recorded from one golf course waterbody. Litoria fallax was the only species to occupy all four waterbody types (Fig. 1).
|
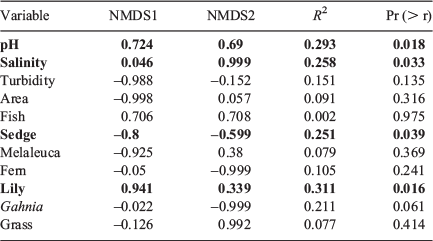
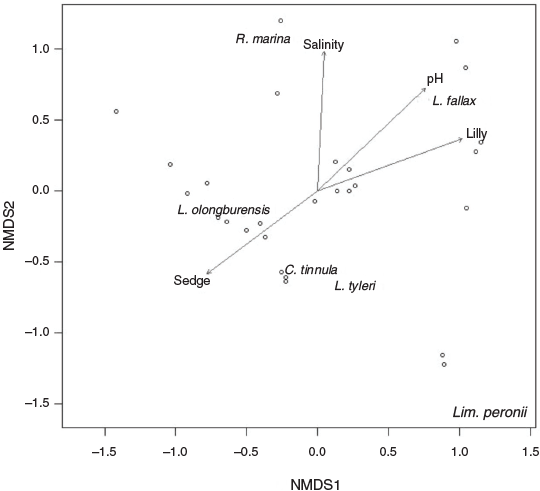
Water pH, salinity, % lily cover and % sedge cover were correlated with change in amphibian assemblage (Table 2). All species were separated on the nMDS plot, with L. olongburensis, C. tinnula and L. tyleri falling within close proximity of each other (Fig. 2).
|
|
Litoria olongburensis and L. fallax relative abundance
Water chemistry variables differed between waterbodies containing L. olongburensis and L. fallax, with pH being higher and sedge cover being lower in waterbodies with L. fallax (Table 1). Waterbody type showed a significant relationship with L. olongburnensis relative abundance (df = 36, F = 3.558, P = 0.025) but not for L. fallax relative abundance (df = 36, F = 1.682, P = 0.19). However, L. fallax was only recorded from two natural waterbodies while the highest abundance of L. olongburensis was recorded from natural waterbodies (Fig. 3). Additionally, no L. olongburensis were recorded from golf courses while the highest relative abundance of L. fallax was recorded from artificial lakes (Fig. 3).
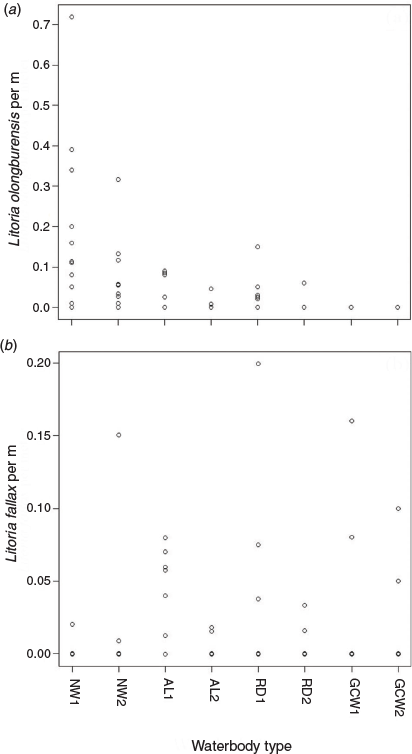
|
Litoria olongburensis were recorded perched on sedge (n = 153), Gahnia sp. (n = 14), Melaleuca sp. (n = 3), fern (n = 4) and grass (n = 2). Litoria fallax were recorded perched on sedges (n = 28), Gahnia sp. (n = 15), lilies (n = 14), Melaleuca sp. (n = 2) and ferns (n = 1).
Tadpoles of L. olongburensis were recorded in 11 natural waterbodies and two roadside ditches, which were adjacent to natural waterbodies. Tadpoles of L. fallax were recorded from one natural waterbody adjacent to a roadside ditch. Tadpoles were only recorded during the summer survey in 2012 despite tadpole surveys being conducted during the 2011 and 2012 surveys.
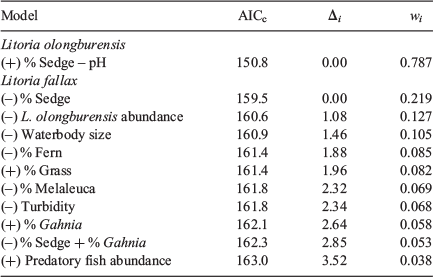
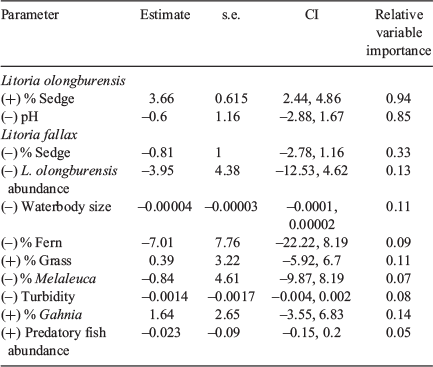
One model for L. olongburensis relative abundance had a Δi ≤ 2 while no models had a Δi ≤ 2–4. The weighting of the best model was 78.7%, indicating that the other models compared poorly (Table 3). This model contained % sedge density and pH. Litoria olongburensis abundance had a positive relationship with increasing % sedge density and a negative relationship with increasing pH. Both % sedge density and pH had high relative variable importance. However, pH had a confidence interval that intersected zero, reducing the evidence that this variable had a strong influence on L. olongburensis relative abundance (Table 4).
|
|
Five models for L. fallax relative abundance had a Δi < 2 and five models had a Δi ≤ 2–4. The combined weighting for models with Δi ≤ 2 was 61.8%. The combined weighting for models with Δi ≤ 2–4 was 28.6%. This gave a total weighting of 90.4% for all models with Δi ≤ 4, indicating that the other models compared poorly (Table 3). Models with a Δi < 2 contained % sedge density, L. olongburensis relative abundance, waterbody size, % fern density and % grass density. All these variables, with the exception of % grass density, were negatively associated with L. fallax relative abundance. Models with a Δi ≤ 2–4 contained % Melaleuca density, % Gahnia density, % sedge density, turbidity and predatory fish. Predatory fish and % Gahnia density were the only variables within models where Δi ≤ 2–4 that were positively associated with L. fallax relative abundance. Percent sedge density had the highest relative variable importance compared with the other variables within models where Δi ≤ 4. However, all variables had a confidence interval that included zero, indicating reduced evidence that these variables had a strong influence on L. fallax relative abundance (Table 4).
Discussion
Frog assemblages
Our results are similar to the majority of surveys conducted within the northern hemisphere (predominantly North America and Europe) that found frog species richness to be higher or equal in artificial, restored waterbodies (reviewed by Brown et al. 2012). Frog assemblage patterns in waterbodies are not consistent, however, and previous Australian studies have reported variable patterns in species richness within and among both natural and constructed waterbodies (Hazell et al. 2004; Lemckert and Mahony 2010; Smallbone et al. 2011; Hamer et al. 2012; Sievers et al. 2018).
The differences and similarities of our findings to previous studies are likely explained by the ability of individual species to respond to differences in the environmental variables that significantly influenced the frog assemblage (i.e. pH, % sedge density, % lily density). Vegetation variables significantly influencing the frog assemblage are likely a result of particular species ‘favouring’ certain varieties of vegetation. This was observed for L. olongburensis, which was found predominantly on sedge species and supports previous studies that found L. olongburensis prefers perching on certain sedge species (Shuker and Hero 2013). Intolerance to water chemistry levels by different frog species tadpoles explains the influence of water chemistry variables on the frog assemblage, as adults have been shown to avoid depositing eggs in waters where water chemistry variables are unfavourable for successful reproduction (Takahashi 2007). For example, post-Gosner stage 25 tadpoles of L. fallax failed to metamorphose when exposed to acidic waters (i.e. pH 3.5), whereas acid-water adapted tadpoles species (i.e. C. tinnula) successfully metamorphosed in pH waters of 3.5, 4.5, and 5.0 (Meyer 2004).
Intolerance to water chemistry variables only explains why some species may be influenced by water chemistry. Species that can tolerate a wide range of water chemistry variable levels may be excluded from or be less common in waterbodies due to competition or predation. It has been hypothesised that competition may explain the distributions of L. fallax and L. olongburensis within waterbodies across the studied landscape (Meyer et al. 2006), as shown by previous studies revealing competitive interactions between other amphibian species (Wiltshire and Bull 1977; Twomey et al. 2008).
Surprisingly, the acid frog species L. freycineti was only recorded from roadside ditches. This species is infrequently encountered in natural waterbodies within coastal wallum systems; however, it is occasionally recorded from disturbed sites (i.e. drainage lines (Meyer et al. 2006), on roads away from wetlands (J.-M. Hero, pers. obs.) and fire trails near water (C. A. Simpkins, pers. obs.). These disturbed areas often have lower vegetation density, which would likely increase detectability of this species when compared with natural waterbodies, where vegetation density is often dense. Therefore, the absence of this species from natural waterbodies may be false and the interpretation of natural habitat usage for this species is difficult. However, our results do indicate that usage of artificial lakes and golf course waterbodies by this species is low, possibly due to higher pH levels and, within golf course waterbodies, competition from the non-acid frog species L. nasuta, which has been proposed as a potential competitor of L. freycineti (Meyer et al. 2006).
Variables influencing L. olongburensis and L. fallax relative abundance
The variables most strongly associated with L. olongburensis relative abundance were high sedge density and low pH. Sedge density positively influenced L. olongburensis abundance with individuals predominantly found perching on sedge species. Amphibians may have coloration that assists with camouflage in their natural environment (Toledo and Haddad 2009). This has been suggested to occur with L. olongburensis, with sedge and dorsal coloration or patterning possibly aiding with camouflage (Lowe and Hero 2012). Our results are consistent with a past survey that showed L. olongburensis were located predominantly within coastal wallum waterbodies containing sedges (Lewis and Goldingay 2005). Our findings are also consistent with Lewis and Goldingay (2005) whose data indicated L. olongburensis are a specialist species predominantly utilising ecological niches with high sedge density and waters where pH < 5.
It is difficult to determine the variables that were influencing L. fallax relative abundance as variables that were within the top models had low relative variable importance and had confidence intervals that included zero. This result suggests that a variable that is strongly influencing L. fallax relative abundance was not measured or that L. fallax is a generalist species, with the variables measured having the same relative influence on L. fallax relative abundance across the natural and modified landscape. Despite this, L. fallax numbers and occupancy within natural waterbodies (where pH was low and sedge density high) were low and were highest within artificial lakes (where pH was high and sedge was intermediate). As mentioned previously, this is likely a result of L. fallax tadpole intolerance to low pH waters (Meyer 2004). Additionally, our results indicate that L. fallax do not require waterbodies where sedge density is high.
Implications for conservation
Differing frog species occurring within different types of waterbodies indicate that a range of waterbody types, both natural and anthropogenic/modified, are required to effectively conserve the full range of assemblages and species currently present within the studied environment. Within wallum heathlands we recommend conservation should be primarily focused on natural waterbodies, where occupancy and presence of tadpoles for threatened, specialist species was highest. Hence, conserving natural waterbodies will be the conservation approach most likely to result in the persistence of the threatened species considered in this study.
The presence of L. olongburensis in artificial lakes and roadside ditches and the presence of C. tinnula in artificial lakes and golf course waterbodies indicate that these waterbodies can provide a form of habitat for adults of wallum associated threatened frog species. These results provide evidence that recommendations to combat the effects of climate change in amphibian populations by establishing artificial waterbodies with longer hydroperiods (Shoo et al. 2011) can be implemented for L. olongburensis and C. tinnula. However, the low number of anthropogenic waterbodies with L. olongburensis tadpoles indicates that the majority of anthropogenic waterbodies are unfavourable for tadpole presence of this species. These waterbodies are likely unsuitable because they have environments with traits that are unfavourable for successful reproduction such as higher pH or incorrect water depths, as outlined by Simpkins et al. (2014). Conversely, while providing potential refuge habitat, these waterbodies may be ecological traps and would not be able to maintain continuing L. olongburensis and, potentially, C. tinnula populations. Furthermore, the road ditches with L. olongburensis tadpoles were adjacent to natural waterbodies. Under these circumstances, road side ditches may be able to permit recruitment as well as connectivity between waterbodies and further study is warranted on this subject.
With the exception of golf course waterbodies, the vast majority of waterbodies surveyed were surrounded by wallum heathland habitat. Therefore, results from this study may only be applicable to waterbodies where wallum heathland habitat is present. One ‘acid’ frog species (L. cooloolensis) has been located up to 1.3 km away from natural waterbodies within undisturbed environments (Simpkins et al. 2011). This suggests dispersal of Litoria ‘acid’ frog species (i.e. L. olongburensis) could occur over equal distances and, provided that natural habitat is still intact, modified/disturbed waterbodies that are established away from natural waterbodies may act as island habitats for adults of this species. Further study, where wallum heathland is not present around modified/disturbed waterbodies, is required to determine if ‘acid’ frogs can utilise waterbodies across disturbed landscapes.
Natural waterbodies where pH and sedge density are within the apparent preferred ranges for L. olongburensis should be given top priority for conservation as these would appear most likely to provide suitable habitat for this species. If construction or conservation of artificial waterbodies is undertaken for habitat loss compensation or in an attempt to reduce the risks of climate change, then the variables found here to be highly correlated with the presence and abundance of L. olongburensis (high sedge density, low pH) should be the target conditions for the waterbody. In addition to this, variables that are ideal for L. olongburensis tadpole presence (Simpkins et al. 2014) and recruitment (Lowe et al. 2015) need to also be attained.
Conflicts of interest
The authors declare no conflicts of interest.
Declaration of funding
Funding for this study was provided by the Griffith School of Environment.
Acknowledgements
The authors would like to thank Evan Pickett and Dianna Virki, who provided valuable comments on drafts of this paper. We would also like to thank Edward Meyer for advice on pH data analysis and sampling techniques. We would like to thank Alan Kerr from the Bribie Island Environmental Protection Association who provided accommodation during fieldwork and Bayshore Resort in Byron Bay, New South Wales, Australia for giving us access to their land. We also thank Chris Touey, Amanda Simpkins, Tempe Parnell, Billy Ross, Matt Davies and Nick Clarke for assisting in data collection.This study was authorised by Griffith Animal Ethics number ENV/28/11/AEC, Queensland Department of Environment and Resource Management ECOACCESS permit number WITK10631812, and New South Wales Department of Environment Scientific License number S21717. The authors dedicate this paper to Dr Phil Bishop, true frog conservationist, who sadly passed in January 2021 and was an assessor on the Master thesis that this paper formed part of.
References
Barry, M. J. (2015). Effects of resource distribution on the cost of predator avoidance behaviour in tadpoles. Hydrobiologia 758, 99–105.| Effects of resource distribution on the cost of predator avoidance behaviour in tadpoles.Crossref | GoogleScholarGoogle Scholar |
Barton, K. (2012). MuMIn: Multi-model inference. R package, ver. 1.7.2. Available at https://cran.r-project.org/
Beebee, T. J. C., and Griffiths, R. A. (2005). The amphibian decline crisis: A watershed for conservation biology? Biological Conservation 125, 271–285.
| The amphibian decline crisis: A watershed for conservation biology?Crossref | GoogleScholarGoogle Scholar |
Brand, A. B., and Snodgrass, J. W. (2010). Value of artificial habitats for amphibian reproduction in altered landscapes. Conservation Biology 24, 295–301.
| Value of artificial habitats for amphibian reproduction in altered landscapes.Crossref | GoogleScholarGoogle Scholar | 19681986PubMed |
Brown, D. J., Street, G. M., Nairn, R. W., and Forstner, M. R. J. (2012). A Place to Call Home: Amphibian Use of Created and Restored Wetlands. International Journal of Ecology 2012, .
| A Place to Call Home: Amphibian Use of Created and Restored Wetlands.Crossref | GoogleScholarGoogle Scholar |
Burnham, K. P., and Anderson, D. R. (2002). ‘Model Selection and Multimodel Inference: A Practical Information-Theoretic Approach.’ (Springer: New York.)
Catterall, C. P. and Kingston, M. (1993). ‘Remnant Bushland of South East Queensland in the 1990’s: Its Distribution, Loss, Ecological Consequences, and Future Prospects.’ (Institute of Applied Environmental Research, Griffith University and Brisbane City Council: Brisbane, Australia.)
Chinathamby, K., Reina, R. D., Bailey, P. C. E., and Lees, B. K. (2006). Effects of salinity on the survival, growth and development of tadpoles of the brown tree frog, Litoria ewingii. Australian Journal of Zoology 54, 97–105.
| Effects of salinity on the survival, growth and development of tadpoles of the brown tree frog, Litoria ewingii.Crossref | GoogleScholarGoogle Scholar |
Christy, M. T., and Dickman, C. R. (2002). Effects of salinity on tadpoles of the green and golden bell frog (Litoria aurea). Amphibia-Reptilia 23, 1–11.
| Effects of salinity on tadpoles of the green and golden bell frog (Litoria aurea).Crossref | GoogleScholarGoogle Scholar |
Collins, J. P., and Storfer, A. (2003). Global amphibian declines: sorting the hypotheses. Diversity and Distributions 9, 89–98.
| Global amphibian declines: sorting the hypotheses.Crossref | GoogleScholarGoogle Scholar |
Crawley, M. J. (2007). ‘The R Book.’ (Wiley: Chichester, West Sussex, United Kingdom)
Filer, A., Beyer, H. L., Meyer, E., and Van Rensburg, B. J. (2019). Distribution mapping of specialized amphbian species in rare, ephemeral habitats: Implications for the conservation of threatened “acid” frogs in south-east Queensland. Conservation Science and Practice 2, .
| Distribution mapping of specialized amphbian species in rare, ephemeral habitats: Implications for the conservation of threatened “acid” frogs in south-east Queensland.Crossref | GoogleScholarGoogle Scholar |
Garden, J. G., McAlpine, C. A., Possingham, H. P., and Jones, D. N. (2007). Using multiple survey methods to detect terrestrial reptiles and mammals: what are the most successful and cost-efficient combinations? Wildlife Research 34, 218–227.
| Using multiple survey methods to detect terrestrial reptiles and mammals: what are the most successful and cost-efficient combinations?Crossref | GoogleScholarGoogle Scholar |
Garden, J. G., McAlpine, C. A., and Possingham, H. P. (2010). Multi-scaled habitat considerations for conserving urban biodiversity: native reptiles and small mammals in Brisbane, Australia. Landscape Ecology 25, 1013–1028.
| Multi-scaled habitat considerations for conserving urban biodiversity: native reptiles and small mammals in Brisbane, Australia.Crossref | GoogleScholarGoogle Scholar |
Griffith, S. J., Bale, C., Adam, P., and Wilson, R. (2003). Wallum and related vegetation on the NSW North Coast: description and phytosociological analysis. Cunninghamia 8, 202–252.
Griffith, S. J., Bale, C., and Adam, P. (2008). Environmental correlates of coastal heathland and allied vegetation. Australian Journal of Botany 56, 512–526.
| Environmental correlates of coastal heathland and allied vegetation.Crossref | GoogleScholarGoogle Scholar |
Grueber, C. E., Nakagawa, S., Laws, R. J., and Jamieson, G. (2011). Multimodel inference in ecology and evolution: challenges and solutions. Journal of Evolutionary Biology 24, 699–711.
| Multimodel inference in ecology and evolution: challenges and solutions.Crossref | GoogleScholarGoogle Scholar | 21272107PubMed |
Hamer, A. J., Smith, P. J., and McDonnell, M. J. (2012). The importance of habitat design and aquatic connectivity in amphibian use of urban stormwater retention ponds. Urban Ecosystems 15, 451–471.
| The importance of habitat design and aquatic connectivity in amphibian use of urban stormwater retention ponds.Crossref | GoogleScholarGoogle Scholar |
Hazell, D., Hero, J.-M., Lindenmayer, D., and Cunningham, R. (2004). A comparison of constructed and natural habitat for frog conservation in an Australian agricultural landscape. Biological Conservation 119, 61–71.
| A comparison of constructed and natural habitat for frog conservation in an Australian agricultural landscape.Crossref | GoogleScholarGoogle Scholar |
Hecnar, S. J., and M’Closkey, R. T. (1996). Amphibian species richness and distribution in relation to pond water chemistry in south-western Ontario, Canada. Freshwater Biology 36, 7–15.
| Amphibian species richness and distribution in relation to pond water chemistry in south-western Ontario, Canada.Crossref | GoogleScholarGoogle Scholar |
Hero, J.-M., Magnusson, W. E., Rocha, C. F. D., and Catterall, C. P. (2001). Antipredator defenses influence the distribution of amphibian prey species in the central Amazon rain forest. Biotropica 33, 131–141.
| Antipredator defenses influence the distribution of amphibian prey species in the central Amazon rain forest.Crossref | GoogleScholarGoogle Scholar |
Heyer, W. R. (1973). Ecological interactions of frog larvae at a seasonal tropical location in Thailand. Journal of Herpetology 7, 337–361.
| Ecological interactions of frog larvae at a seasonal tropical location in Thailand.Crossref | GoogleScholarGoogle Scholar |
Hines, H., and Meyer, E. (2011). The frog fauna of Bribie Island:an annoted list and comparison with other comparison with other Queensland dune islands. Proceedings of the Royal Scoiety of Queensland 117, 261–274.
Hines, H. B., Mahony, M., and McDonald, K. (1999). An assessment of frog declines in wet subtropical Australia. In ‘Decline and disappearances of Australian Frogs’. (Ed. A. Campbell.) pp. 44–63. (Environment Australia: Canberra.)
Ingram, G. J., and Corben, C. J. (1975). The frog fauna of North Stradbroke Island, with comments on the ‘acid’ frogs of the wallum. The Proceedings of the Royal Society of Queensland 86, 49–54.
Johnson, J. B., and Omland, K. S. (2004). Model selection in ecology and evolution. Trends in Ecology & Evolution 19, 101–108.
| Model selection in ecology and evolution.Crossref | GoogleScholarGoogle Scholar |
Kats, L. B., Petranka, J. W., and Sih, A. (1988). Antipredator defenses and the persistence of amphibian larvae with fishes. Ecology 69, 1865–1870.
| Antipredator defenses and the persistence of amphibian larvae with fishes.Crossref | GoogleScholarGoogle Scholar |
Lemckert, F., and Mahony, M. (2010). The relationship amoung multiple-scale habitat variables and pond use by anruans in northern New South Wales, Australia. Herpetological Conservation and Biology 5, 537–547.
Levin, N., Elron, E., and Gasith, A. (2009). Decline of wetland ecosystems in the coastal plain of Israel during the 20th century: Implications for wetland conservation and management. Landscape and Urban Planning 92, 220–232.
| Decline of wetland ecosystems in the coastal plain of Israel during the 20th century: Implications for wetland conservation and management.Crossref | GoogleScholarGoogle Scholar |
Lewis, B. D., and Goldingay, R. L. (2005). Population monitoring of the vulnerable wallum sedge frog (Litoria olongburensis) in north-eastern New South Wales. Australian Journal of Zoology 53, 185–194.
| Population monitoring of the vulnerable wallum sedge frog (Litoria olongburensis) in north-eastern New South Wales.Crossref | GoogleScholarGoogle Scholar |
Lowe, K., and Hero, J.-M. (2012). Sexual Dimorphism and Color Polymorphism in the Wallum Sedge Frog (Litoria olongburensis). Herpetological Review 43, 236–240.
Lowe, K., Castley, J. G., and Hero, J.-M. (2015). Resilience to climate change: complex relationships among wetland hydroperiod, larval amphibians and aquatic predators in temporary wetlands. Marine and Freshwater Research 66, 886–899.
| Resilience to climate change: complex relationships among wetland hydroperiod, larval amphibians and aquatic predators in temporary wetlands.Crossref | GoogleScholarGoogle Scholar |
Magnus, R., and Rannap, R. (2019). Pond construction for threatened amphibians is an important conservation tool, even in landscapes with extant natural water bodies. Wetlands Ecology and Management 27, 323–341.
| Pond construction for threatened amphibians is an important conservation tool, even in landscapes with extant natural water bodies.Crossref | GoogleScholarGoogle Scholar |
Mazerolle, M. J., Poulin, M., Lavoie, C., Rochefort, L., Desrochers, A., and Drolet, B. (2006). Animal and vegetation patterns in natural and man-made bog pools: implications for restoration. Freshwater Biology 51, 333–350.
| Animal and vegetation patterns in natural and man-made bog pools: implications for restoration.Crossref | GoogleScholarGoogle Scholar |
Meyer, E. (2004). Acid adaptation and mechanisms for softwater acid tolerance in larvae of anuran species native to the ‘Wallum’ of east Australia. PhD thesis, University of Queensland, Brisbane.
Meyer, E., Hero, J.-M., Shoo, L., and Lewis, B. (2006). National Recovery Plan for the Wallum Sedgefrog and Other Wallum-Dependent Frog Species. Report to Department of the Environment and Water Resources, Canberra. Queensland Parks and Wildlife Service, Brisbane.
Mokany, A., and Shine, R. (2003). Competition between tadpoles and mosquito larvae. Oecologia 135, 615–620.
| Competition between tadpoles and mosquito larvae.Crossref | GoogleScholarGoogle Scholar | 12684864PubMed |
Monello, R. J., and Wright, R. G. (1999). Amphibian habitat preferences among artificial ponds in the Palouse region of northern Idaho. Journal of Herpetology 33, 298–303.
| Amphibian habitat preferences among artificial ponds in the Palouse region of northern Idaho.Crossref | GoogleScholarGoogle Scholar |
Morin, P. J. (1986). Interactions Between Intraspecific Competition and Predation in an Amphibian Predator-Prey System. Ecology 67, 713–720.
| Interactions Between Intraspecific Competition and Predation in an Amphibian Predator-Prey System.Crossref | GoogleScholarGoogle Scholar |
Nowakowski, A. J., Thompson, M. E., Donnelly, M. A., and Todd, B. D. (2017). Amphibian sensitivity to habitat modification is associated with population trends and species traits. Global Ecology and Biogeography 26, 700–712.
| Amphibian sensitivity to habitat modification is associated with population trends and species traits.Crossref | GoogleScholarGoogle Scholar |
Oksanen, J. (2011). Multivariate analysis of ecological communities in R: vegan tutorial. Available at https://john-quensen.com/wp-content/uploads/2018/10/Oksanen-Jari-vegantutor.pdf [Verified 20 September 2012]
Oksanen, J., Blanchet, F. G., Kindt, R., Legendre, P., Minchin, P. R., O’Hara, R. B., Simpson, G. L., Solymos, P., Stevens, M. H. H., and Wagner, H. (2012). Vegan: Community Ecology Package, ver. 2.0-4. Available at http://vegan.r-forge.r-project.org/.
Pierce, B. A. (1985). Acid Tolerance in Amphibians. BioScience 35, 239–243.
| Acid Tolerance in Amphibians.Crossref | GoogleScholarGoogle Scholar |
Pyron, R. A. (2018). Global amphibian declines have winners and losers. Proceedings of the National Academy of Science 115, 3739–3741.
| Global amphibian declines have winners and losers.Crossref | GoogleScholarGoogle Scholar |
R Core Development Team (2011). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria.
Rannap, R., Lõhmus, A., and Briggs, L. (2009). Restoring ponds for amphibians: a success story. Hydrobiologia 634, 87–95.
| Restoring ponds for amphibians: a success story.Crossref | GoogleScholarGoogle Scholar |
Rios-López, N. (2008). Effects of increased salinity on tadpoles of two anurans from a Caribbean coastal wetland in relation to their natural abundance. Amphibia-Reptilia 29, 7–18.
| Effects of increased salinity on tadpoles of two anurans from a Caribbean coastal wetland in relation to their natural abundance.Crossref | GoogleScholarGoogle Scholar |
Robertson, B. A., and Hutto, R. L. (2006). A framework for understanding ecological traps and an evaluation of existing evidence. Ecology 87, 1075–1085.
| A framework for understanding ecological traps and an evaluation of existing evidence.Crossref | GoogleScholarGoogle Scholar | 16761584PubMed |
Ruhí, A., Sebastian, O. S., Feo, C., Franch, M., Gascón, S., Richter-Boix, À., Boix, D., and Llorente, G. (2012). Man-made Mediterranean temporary ponds as a tool for amphibian conservation. International Journal of Limnology 48, 81–93.
| Man-made Mediterranean temporary ponds as a tool for amphibian conservation.Crossref | GoogleScholarGoogle Scholar |
Shoo, L. P., Olson, D. H., McMenamin, S. K., Murray, K. A., Van Sluys, M., Donnelly, M. A., Stratford, D., Terhivuo, J., Merino-Viteri, A., Herbert, S. M., Bishop, P. J., Corn, P. S., Dovey, L., Griffiths, R. A., Lowe, K., Mahony, M., McCallum, H., Shuker, J. D., Simpkins, C., Skerratt, L. F., Williams, S. E., and Hero, J.-M. (2011). Engineering a future for amphibians under climate change. Journal of Applied Ecology 48, 487–492.
| Engineering a future for amphibians under climate change.Crossref | GoogleScholarGoogle Scholar |
Shuker, J. D., and Hero, J.-M. (2013). Perch substrate use by the threatened wallum sedge frog (Litoria olongburensis) in wetland habitats of mainland eastern Australia. Australian Journal of Zoology 60, 219–224.
| Perch substrate use by the threatened wallum sedge frog (Litoria olongburensis) in wetland habitats of mainland eastern Australia.Crossref | GoogleScholarGoogle Scholar |
Shuker, J. D., Simpkins, C. A., and Hero, J. M. (2016). Determining environmental limits of threatened species: The example of the wallum sedgefrog Litoria olongburensis. Ecosphere 7, 1–13.
| Determining environmental limits of threatened species: The example of the wallum sedgefrog Litoria olongburensis.Crossref | GoogleScholarGoogle Scholar |
Sievers, M., Hale, R., Swearer, S. E., and Parris, K. M. (2018). Frog occupancy of polluted wetlands in urban landscapes. Conservation Biology 33, 389–402.
| Frog occupancy of polluted wetlands in urban landscapes.Crossref | GoogleScholarGoogle Scholar | 30151963PubMed |
Simpkins, C. A. (2012). Aboitic and biotic factors influencing the assemblage of tadploes and adult anurans in coastal wallum habitats of eastern Australia. Masters thesis, Griffith University, Brisbane.
Simpkins, C. A., Meyer, E., and Hero, J.-M. (2011). Long-range movement in the rare Cooloola sedgefrog Litoria cooloolensis. Australian Zoologist 35, 977–978.
| Long-range movement in the rare Cooloola sedgefrog Litoria cooloolensis.Crossref | GoogleScholarGoogle Scholar |
Simpkins, C. A., Shuker, J. D., Lollback, G. W., Castley, J. G., and Hero, J.-M. (2014). Environmental variables associated with the distribution and occupancy of habitat specialist tadpoles in naturally acidic, oligotrophic waterbodies. Austral Ecology 39, .
| Environmental variables associated with the distribution and occupancy of habitat specialist tadpoles in naturally acidic, oligotrophic waterbodies.Crossref | GoogleScholarGoogle Scholar |
Smallbone, L., Luck, G. W., and Wassens, S. (2011). Anuran species in urban landscapes: Relationships with biophysical, built environment and socio-economic factors. Landscape and Urban Planning 101, 43–51.
| Anuran species in urban landscapes: Relationships with biophysical, built environment and socio-economic factors.Crossref | GoogleScholarGoogle Scholar |
Sparling, D. W. (2010). Water-quality criteria for amphibians. In ‘Amphibian Ecology and Conservation: A Handbook of Techniques’. (Ed. C. K. Dodd, Jr.). pp 105–120. (Oxford University Press: New York.)
Strahan, R. (1957). The Effect of Salinity on the Survival of Larvae of Bufo melanostictus Schneider. Copeia 1957, 146–147.
| The Effect of Salinity on the Survival of Larvae of Bufo melanostictus Schneider.Crossref | GoogleScholarGoogle Scholar |
Stuart, S. N., Chanson, J. S., Cox, N. A., Young, B. E., Rodrigues, A. S. L., Fischman, D. L., and Waller, R. W. (2004). Status and trends of amphibian declines and extinctions worldwide. Science 306, 1783–6.
| Status and trends of amphibian declines and extinctions worldwide.Crossref | GoogleScholarGoogle Scholar | 15486254PubMed |
Takahashi, M. (2007). Oviposition site selection: pesticide avoidance by gray treefrogs. Environmental Toxicology and Chemistry 26, 1476–1480.
| 17665689PubMed |
Toledo, L. F., and Haddad, C. F. B. (2009). Colors and some morphological traits as defensive mechanisms in anurans. International Journal of Zoology 2009, 1–12.
| Colors and some morphological traits as defensive mechanisms in anurans.Crossref | GoogleScholarGoogle Scholar |
Turner, R. E. (1990). Landscape Development and Coastal Wetland Losses in the Northern Gulf of Mexico. American Zoologist 30, 89–105.
| Landscape Development and Coastal Wetland Losses in the Northern Gulf of Mexico.Crossref | GoogleScholarGoogle Scholar |
Twomey, E., Morales, V., and Summers, K. (2008). Evaluating condition-specific and asymmetric competition in a species-distribution context. Oikos 117, 1175–1184.
| Evaluating condition-specific and asymmetric competition in a species-distribution context.Crossref | GoogleScholarGoogle Scholar |
Vonesh, J. R., Kraus, J. M., Rosenberg, J. S., and Chase, J. M. (2009). Predator effects on aquatic community assembly: disentangling the roles of habitat selection and post-colonization processes. Oikos 118, 1219–1229.
| Predator effects on aquatic community assembly: disentangling the roles of habitat selection and post-colonization processes.Crossref | GoogleScholarGoogle Scholar |
Wilbur, H. M. (1987). Regulation of structure in complex systems: experimental temporary pond communities. Ecology 68, 1437–1452.
| Regulation of structure in complex systems: experimental temporary pond communities.Crossref | GoogleScholarGoogle Scholar |
Wiltshire, D. J., and Bull, C. M. (1977). Potential competitive interactions between larvae of Pseudophryne bibroni and P. semimarmorata (Anura: Leptodactylidae). Australian Journal of Zoology 25, 449–454.
| Potential competitive interactions between larvae of Pseudophryne bibroni and P. semimarmorata (Anura: Leptodactylidae).Crossref | GoogleScholarGoogle Scholar |


