The Pacific foodfish list: a standardized list of over 1000 species of marine fish consumed by Pacific people
N. L. Andrew A * , L. Aisea A B , N. D. H. Arachchi
A * , L. Aisea A B , N. D. H. Arachchi  A , N. Bailly C D , I. Bertram E , P. Bosserelle F , L. Carron G , K. D. Clements
A , N. Bailly C D , I. Bertram E , P. Bosserelle F , L. Carron G , K. D. Clements  H , M. P. Duenas I , V. Fatongiatau B , S. Felise J , S. González-Murcia K , B. Jaugeon L , L. Joy M , L. P. Kaitu N , U. Kaly O , J.-A. Kerandel E , J. Z. Koehn P , J. Kinch Q , S. Jalam R , A. Kwapena S , O. Li A , S. Lindfield T , F. Magron E , B. R. Moore U , R. F. Myers V , B. Ned W , B. Nikiari X , D. Ochavillo J , J. van der Ploeg
H , M. P. Duenas I , V. Fatongiatau B , S. Felise J , S. González-Murcia K , B. Jaugeon L , L. Joy M , L. P. Kaitu N , U. Kaly O , J.-A. Kerandel E , J. Z. Koehn P , J. Kinch Q , S. Jalam R , A. Kwapena S , O. Li A , S. Lindfield T , F. Magron E , B. R. Moore U , R. F. Myers V , B. Ned W , B. Nikiari X , D. Ochavillo J , J. van der Ploeg  Y , K. Raumea Z , M. Roscher A , M. Sabater AA , A. Sami L , G. Shedrawi A E , N. Smith E , D. J. Steenbergen
Y , K. Raumea Z , M. Roscher A , M. Sabater AA , A. Sami L , G. Shedrawi A E , N. Smith E , D. J. Steenbergen  A , M. Sukulu AB , F. Tulafono AC , S. Tiitii AD , T. Toroca AE , P. Tua AF , L. Vaipuna B , C. C. C. Wabnitz P , J. Wencélius AG , M. White AH and B. Yeeting AI
A , M. Sukulu AB , F. Tulafono AC , S. Tiitii AD , T. Toroca AE , P. Tua AF , L. Vaipuna B , C. C. C. Wabnitz P , J. Wencélius AG , M. White AH and B. Yeeting AI
A
B
C
D
E
F
G
H
I
J
K
L
M
N
O
P
Q
R
S
T
U
V
W
X
Y
Z
AA
AB
AC
AD
AE
AF
AG
AH
AI
Abstract
The sustainable supply of fish in the face of climate change and other drivers of change is a policy priority for Pacific nations. Creel and market surveys are increasingly used to document catches but this information has not been aggregated at a regional scale.
In this paper we provide a comprehensive and standardized list of Pacific marine foodfishes in 22 Pacific Island Countries and Territories to enable comparative analyses and improved national surveys.
National lists of marine teleost fish caught for food were cleaned of errors and standardized to current valid names using authoritative global databases. National lists were subsequently aggregated to subregional and regional scales.
Pacific people were found to consume more than 1000 species of marine fish (presently 1031 species), with the highest diversity observed in Melanesia and the lowest in Polynesia. A total of 14% of species names have changed since surveys were completed. An estimated 3847 species of marine fish are recorded from the region, most of which are small reef species. This list of Pacific foodfish is available through the Pacific Data Hub curated by the Pacific Community.
The study quantifies, for the first time, the great diversity of fishes consumed by Pacific people and highlights the need for more baselines of catch, acquisition and consumption.
These findings provide a foundation across the region for analysing species’ relative importance in local economies and diets, supporting fisheries management and food security policies critical to the wellbeing of Pacific people in a changing world.
Keywords: creel surveys, English common names, fish catch, fish species diversity, foodfish, market surveys, Pacific region, pelagic fish, PICTs, reef fish.
Introduction
Catching, trading and eating fish is central to the Pacific way of life. Most catch comes from reefs, lagoons and mangrove forests, and fish are among the most accessible and widely consumed animal source food for coastal communities (Gillett and Fong 2023). The sustainable supply of coastal fish has been of concern in the technical literature for some time (e.g. Bell et al. 2009) and there is growing evidence that the status quo in coastal fisheries will lead to inadequate supplies of fish in the face of human population growth and climate change. The cost of replacing the food provision and employment benefits provided by these fisheries would place huge demands on the environment and national economies. This ‘fish supply gap’ (Bell et al. 2009, 2015, 2018) has gained renewed prominence in policy and political domains with the 2014 recognition of coastal fisheries as one of six regional priorities within the Framework for Pacific Regionalism (https://www.adb.org/sites/default/files/linked-documents/pacific-robp-2015-2017-sd.pdf).
The great importance of fish in the lives of Pacific people reflects the dominance of marine ecosystems in the region. Marine areas of the Pacific Islands region encompass over 27 million km2, comprising 98% of the total area under the jurisdiction of Pacific Island Countries and Territories (PICTs). The region has over 140,000 km2 of coral reefs (Hoegh-Guldberg et al. 2011), along with extensive lagoonal, nearshore and open ocean waters. These habitats support an extraordinary diversity of fish, with over 4100 native marine species reported from the region (Froese and Pauly 2000) and likely many more still unknown to science. At a regional scale, fish species diversity decreases from west to east (Parravicini et al. 2013), with the greatest diversity occurring in Papua New Guinea and Solomon Islands which form part of what has come to be known as the Coral Triangle, the most biologically diverse marine region in the world (Hoeksema 2007; Veron et al. 2011; Edgar et al. 2014). At a local scale, for reef-associated fish, diversity varies with several factors including reef type, area and complexity (Bellwood et al. 2005; Yeager et al. 2017), wave exposure/lagoon flushing (Houk et al. 2012) and depth (Friedlander et al. 2010), and the degree of human impact (e.g. fishing and habitat modification) (Goetze et al. 2011; D’agata et al. 2014).
Pacific people are known to be among the world’s highest consumers of fish (FAO 2020; Gillett and Fong 2023). Nevertheless, the region is not uniform and while some Micronesian atoll countries consume more than 100 kg per capita year−1, consumption in others is comparable to the global average of 19 kg per capita year−1 (Sharp and Andrew 2024). Given this prominence in diets, local economies and culture, how little is known about the production, acquisition and consumption of fish in this region is perhaps surprising. Importantly, also, the assumption that the region is homogenous in its use of fish will not promote the contextualized interventions needed to maximize contributions to food and nutrition security. There are, for example, few reliable descriptions of the types of fish eaten, by whom and where. There are many reasons for the lack of information about the production, trade and consumption of fish, including the dispersed and remote nature of many fisheries and under-resourced national agencies. These constraints are compounded by the intrinsic diversity of fisheries and the enormous complexity of the underlying ecological characteristics of these resources.
We provide a comprehensive, standardized list of Pacific foodfish. We use the closed form of the compound noun ‘foodfish’ for brevity and in recognition of common usage of the term. Such a standardized list, publicly available and held current by The Pacific Community (SPC), will provide an essential reference for comparative analyses of patterns and trends in the types and relative importance of fish consumed in the region. The list will further enable analyses of changes in fish catches under climate change and a range of research on the social dimensions of fisheries. Adams and Dalzell (1999), in a review of Pacific coastal fisheries, remarked that a ‘creel surveyor would need to identify at least one hundred species of finfish’. In the 26 years since that time, the number of creel and market surveys has increased significantly but this information has not been aggregated and updated at a regional scale.
In simplest terms, a ‘foodfish’ may be any fish that people eat but this potentially includes all fish found across the region and therefore provides no pragmatic foundation for improved analysis in domains ranging from fisheries management, codification of local names, and prioritising the collection of nutrient composition profiles of target species. We define a foodfish as any species recorded by national agencies and partners as being caught for food. Our focus is on teleost foodfish – subsequent lists will include elasmobranchs and invertebrates. In practice, foodfish may be surveyed under a wide range of circumstances, including at landing sites in communities, places where catches are aggregated for distribution within local and national values chains and in markets. In this context, a ‘market’ may range from a roadside stall to a regulated formal market in a capital city (Fig. 1). These survey locations reflect the diverse ways in which Pacific people acquire fish, some of which may involve a cash transaction but also from home production, bartering, gifting and institutional sources such as schools and churches (Bogard et al. 2021).
In addition to catching fish themselves, Pacific Islanders acquire fish from a range of sources including family and at cultural occasions, and also from a diverse range of formal and informal markets. Surveys may intercept catches at, for example, landing sites, roadside stalls and in central markets in major urban centers. Photo location and credits clockwise from top left: Tarawa, Kiribati (Dirk Steenbergen); Papara, Tahiti (Pauline Bosserelle); Uripiv, Vanuatu (Eleanor McNeill); Taravao, Tahiti (Pauline Bosserelle); Auki market, Solomon Islands (Filip Milovac); and Papeete, Tahiti (CPS Angèle Armando).

Materials and methods
Geographic and taxonomic scope
Our analysis is restricted to the 22 PICTs that are members of the Pacific Community (SPC; hereafter, ‘Pacific region’ and ‘the region’ refer to those PICTs (Fig. 2). This geographic frame excludes some places in the Pacific region, such as Rapa Nui, Hawai‘i and the Minor Outlying Islands of the United States of America.
Geographic location and extent of Pacific Island Country and Territory Exclusive Economic zones (EEZ) categorized by subregions. Three digit ISO codes are shown for each PICT as: American Samoa (ASM), Cook Islands (COK), Federated States of Micronesia (FSM), Fiji (FJI), French Polynesia (PYF), Guam (GUM), Kiribati (KIR), Marshall Islands (MHL), Nauru (NRU), New Caledonia (NCL), Niue (NIU), Northern Mariana Islands (MNP), Palau (PLW), Papua New Guinea (PNG), Pitcairn Islands (PCN), Samoa (WSM), Solomon Islands (SLB), Tokelau (TKL), Tonga (TON), Tuvalu (TUV), Vanuatu (VUT), and Wallis and Futuna (WLF). Red dots indicate islands or provinces in which catch surveys were done (see Table S2 for a list of islands). Scale bar, survey locations and extent of EEZs are indicative only.

To further circumscribe the task, the list was confined to native marine bony fish (Infraclass Teleostei) found in shallow (<200 m depth) waters in the Exclusive Economic Zones of the 22 PICTs of the region (Fig. 2). Consequently, the following kinds of fish were excluded: (i) species that spend their lives exclusively or mostly in freshwater (notably genus Anguilla), (ii) sharks, rays and chimaeras (class Chondrichthyes), (iii) teleost species categorized as bathydemersal or bathypelagic in FishBase (Froese and Pauly 2000), and those recorded as occurring in water deeper than ~200 m, (iv) introduced fish such as Tilapia (Oreochromis spp.) and (v) imported frozen or chilled fish product (e.g. Oncorhynchus spp.). Given the nature of the fisheries surveyed, exclusion of deepwater species meant, in practice, that only 12 species recorded in national surveys were excluded, along with 23 freshwater/euryhaline species (Supplementary Table S1).
Sources of species lists
Lists of species recorded in national surveys of catches at landing sites and markets were drawn from: (i) published sources, (ii) unpublished lists from national agencies and partners, (iii) the SPC Pacific Regional Oceanic and Coastal Fisheries Development (PROCFish/C/CoFish) program that collected information from 17 PICTs between 2002 and 2009 (Pinca et al. 2010), (iv) unpublished lists from the SPC-led creel and market surveys since 1997, including the Tails and Tails+ program implemented in collaboration with 15 PICTs, (v) community sampling implemented by national agencies in Kiribati and Vanuatu within the Pathways program (Andrew et al. 2020; Campbell et al. 2024), (vi) community and market sampling by WorldFish in Malaita Province in the Solomon Islands (Sulu et al. 2018; Smallhorn-West et al. 2022) and (vii) from the unpublished SPC-led ‘Ikasavea’ program implemented in collaboration with PICTs (Shedrawi et al. 2024). Information from these sources was augmented by experts from national agencies, with particular reference to coastal foodfish of the region listed in Moore and Colas (2016). A complete summary of sources for each PICT is provided in Table S2. The surveys were implemented in a broad range of islands and provinces across the region (Fig. 2, Table S2).
Lists of species from the aquarium trade were not sourced but species that may occur in such lists were included if reported in creel or market surveys. Species recognized as foodfish but protected by national regulation (e.g. Humphead Wrasse, Cheilinus undulatus, in New Caledonia since 2008) were included. A total of 67 species in lists from American Samoa, Guam and Northern Mariana Islands were recorded from single catches from a single PICT in a subregion. To preserve fisher confidentiality, these species are reported only at regional level.
Curating the Pacific foodfish list
National lists of scientific and English common names were checked and standardized with reference to authoritative global lists:
FishBase (Froese and Pauly 2000), electronic version initially accessed online as ‘version 6 June 2023’ and revised in February 2025;
Eschmeyer’s Catalog of Fishes (Fricke et al. 2025), hereafter ‘ECoF’, electronic version initially accessed online as ‘version 2 October 2023’ and revised 2 February 2025;
UN FAO Aquatic Sciences and Fisheries Information System (ASFIS) list of species of fishery and commercial interest (Garibaldi and Busilacchi 2002; FAO 2024), hereafter ‘FAO’. The 2024 version of ASFIS was accessed online as version 14 November 2024; and
The 8th edition of the American Fisheries Society list of English common and scientific names of fishes from the United States, Canada and Mexico (Page et al. 2023), hereafter ‘AmFS’.
These lists have different purposes, geographic coverage, limitations and advantages. Unsurprisingly, they do not always agree. The FAO list is the only international list of English common names produced by a United Nations agency and is focused on species with fishery production statistics. FishBase and ECoF are international lists inclusive of all fishes but the latter does not include English common names. The AmFS list of English common names is produced by a learned society to provide a comprehensive list of all fish species in Canada, the United States and Mexico. The AmFS list explicitly excludes Hawai‘i and by inference American Pacific Territories. These compilations are not independent – FAO cites FishBase and ECoF as sources, for example and all refer the reader to original sources for taxonomy and distributions.
Scientific names
Scientific names reported in surveys were revised to currently accepted species names following FishBase and ECoF. These sources agreed in the great majority of cases but ECoF was used where they did not agree. Family group scientific names follow van der Laan et al. (2014) and as reported by Fricke et al. (2023). Changes to scientific names reported in the original surveys are listed in the ‘Pacific foodfish list’ available at the Pacific Data Hub curated by SPC (https://pacificdata.org/). Note that the listed synonyms are only those reported in the original surveys and not a comprehensive list of synonyms for that species. Only named species are included.
Given their prominence in Pacific fisheries, two taxonomic tribes were separated from their families. The scombrid tribe Thunnini was separated as ‘tuna’; all other tribes in the family (Scombrini, Scomberomorini, and Sardini) are reported as Scombridae. In Family Labridae, the eight genera of parrotfish were separated as Tribe Scarini and all other tribes (Hypsigenyini, Cirrhilabrini, Labrini, Cheilinini, Novaculini and Pseudolabrini) reported as Family Labridae.
Species occurrences and habitat associations
Records in national surveys were assumed to be correct and retained unless the species was not reported from the subregion by ECoF, FishBase or supplementary sources. Distributions of species from FishBase and ECoF were cross-checked with checklists of species found in the region: Allen and Munday (1995), Allen and Erdmann (2012), Dalzell and Preston (1992), Fricke et al. (2023), Friedlander et al. (2014), Hubert et al. (2017), Kulbicki et al. (2011), Laboute and Grandperrin (2000), McKenna et al. (2015), Myers (1999), Myers et al. (2025), Parenti (2021), Randall (1999, 2005), Randall et al. (1997, 2004), Seeto and Baldwin (2010), Siu et al. (2017) and Wright and Hill (1993). These sources were augmented by: (i) those cited in Table S2, (ii) an unpublished checklist of fishes from 18 PICTs curated by Robert Myers (R. F. Myers, unpubl. data) and (iii) for PNG by an unpublished checklist of reef fishes from Kimbe Bay in the Bismarck Sea developed by Geoff Jones and colleagues (G. P. Jones, unpubl. data).
On the western margin of the Pacific region, Indonesia, Philippines and Japan are geographically close to Federated States of Micronesia, Guam, Northern Mariana Islands, Palau and Papua New Guinea (PNG) (Fig. 2). Use of phrases in ECoF and FishBase such as ‘Indo-Pacific’ or ‘east to Philippines’ without reference to a PICT was not taken to mean the species occurred in a PICT and the record was not included.
A total of 261 species was recorded in national lists but not reported in FishBase, ECoF or supplementary sources as occurring within the region (Table S1). Prior to exclusion, these species were further checked against the Global Biodiversity Information Facility database (GBIF 2023, accessed 18 December 2023). In most cases, distributions reported in all three sources were in accord as not recording the species from the region and the species was excluded. In 50 cases, FishBase, ECoF and GBIF were in conflict but after review and replacements caused by name changes and other taxonomic revisions were accounted for, only two species, Halichoeres marginatus and H. zeylonicus were added.
In some cases, species names could reasonably be replaced, such as when previously valid species were split on a geographic basis and could be replaced with a sister species (e.g. Acanthopagrus berda to A. pacificus (Iwatsuki et al. 2010) and Lampris guttatus to L. megalopsis (Underkoffler et al. 2018)) or when an error appeared that could reasonably be corrected, such as reporting Atlantic Bluefin Tuna instead of Pacific Bluefin Tuna. In almost all instances, FishBase and ECoF agreed on broad geographic distributions and were used in combination but where not, ECoF was prioritized (see Table S1 for a list of species replaced).
FishBase habitat descriptions were used as the basis of categorizing habitat associations of species. Categories relevant to this study were pelagic, pelagic-neritic, pelagic-oceanic, benthopelagic, demersal and reef associated. For the purposes of this analysis the first four categories listed above were collapsed into ‘pelagic’. Demersal and reef associated were combined as ‘reef’ species. Given the diversity of benthic marine habitats, from coral reefs to mangrove forests and sandy lagoons, the ‘reef’ category should be interpreted as a generic category encompassing all benthic habitats.
In addition to being appropriate to the broad-brush purpose of this analysis, the ‘pelagic’, ‘reef’ and ‘tuna’ categories were chosen to align with those used in national household surveys of food acquisition. In household surveys, respondents are asked to recall acquisition of fresh and frozen fish by these three categories as well as ‘canned’ and ‘processed’ fish (dried and smoked). National household surveys are the primary tool for estimating the acquisition and consumption of fish in the region (Sharp and Andrew 2021).
Comparison with regional biodiversity
The representativeness of the foodfish list was assessed by comparison with an aggregated list of fish known to occur in the region. Source lists were combined from the sources listed above and filtered using the same criteria used to curate the foodfish list. As for the foodfish list, inclusion of species found in the region necessitated judgements as to whether these met the criteria used to bound the Pacific foodfish list. Inevitably, at the margins we will have excluded euryhaline and deepwater species that other authors may have included. With the possible exception of gobioid fishes (Parenti 2021), such species are not a large proportion of the species pool and their inclusion, or not, would not be consequential to the broad comparisons made. The resulting list is not an authoritative checklist of species names or occurrences by subregion and is used only for broad comparison of the proportions of different kinds of fish captured in the foodfish list. These sources and the broader scientific literature can be consulted for authoritative descriptions of species distributions.
English common names and eponyms
Common names were allocated from FAO and if not on that list, then from, in order: AmFS, Moore and Colas (2016), an unpublished list curated by SPC, and FishBase. Sources of common names were heavily cross-referenced among sources, with all using names aggregated from other sources. As will be seen below, this leads to almost complete congruence among these sources.
English common names were considered the same if they differed only in the form of the compound modifier used to describe the species. To illustrate this, the following names of a hypothetical triggerfish would be considered the same: onespot, one-spot, one spot, one-spotted and one spotted triggerfish. Similarly, minor differences in spelling (e.g. color vs colour or sweetlip vs sweetlips) were not considered different names. Consistent with Page et al.’s (2023) principle concerning simplicity in names, hyphens were removed from English common names where appropriate. Eleven of the 1031 species in the foodfish list have not been assigned a common name in any source and were excluded from the analysis of common names. No new names were coined in the course of the analysis.
The standardized list of scientific and English common names of Pacific foodfish was searched for eponyms in either genus or species names. The etymology of scientific names honoring a person or institution was sourced from the ETYFish database (Scharpf 2024; accessed 6 January 2025).
Sizes of fish
To compare the relative sizes of fish species caught among PICTs and to assess the comprehensiveness of surveys by size and taxonomy, we downloaded estimates of maximum length from FishBase (Froese and Pauly 2000). Maximum length (Lmax) was reported as the longest length reported for a species and should not be interpreted as asymptotic length (Linf). Only a relatively small proportion of species in FishBase have a Linf reported. All reported lengths (as standard or fork) were converted to total length using the conversions reported in FishBase (Binohlan et al. 2011). Where there were sex related differences in Lmax, the larger of the two lengths is reported. If conversions were unavailable at the species level, the mean conversion ratio for species at the genus level or, in a few cases where this was not possible, mean ratios at the family level were used to estimate total lengths. Following these steps, we were able to calculate Lmax for all except 171 species. We acknowledge the limitations of using reported Lmax as a metric of the sizes of fish but contend that this provides an adequate basis for the broad comparisons made. For the subset of species in FishBase that report both Linf and Lmax, the latter was, on average, ca 20% less than Linf.
Results
Pacific foodfish
Across the region, a total of 1031 species of marine fish was recorded as foodfish from 120 families (see ‘Pacific foodfish list’ available on Pacific Data Hub). Most foodfish species (82%) were reef-associated, followed by pelagic species (17%), with tuna accounting for only 1% of species. Consistent with patterns in the biogeography of fish in the region, the greatest diversity of fish species was recorded in catches in Melanesia (818 species), followed by Micronesia (674) and Polynesia (525) (Fig. 3a). A total of 415 widely distributed foodfish species were common to all three subregions.
Overlap in species occurrence among subregions from: (a) creel and market surveys and (b) occurrences in the Pacific region. Numbers indicate the counts of species common to the subregional grouping. Areas of circles and overlap are proportional to species counts for the combination of subregions.

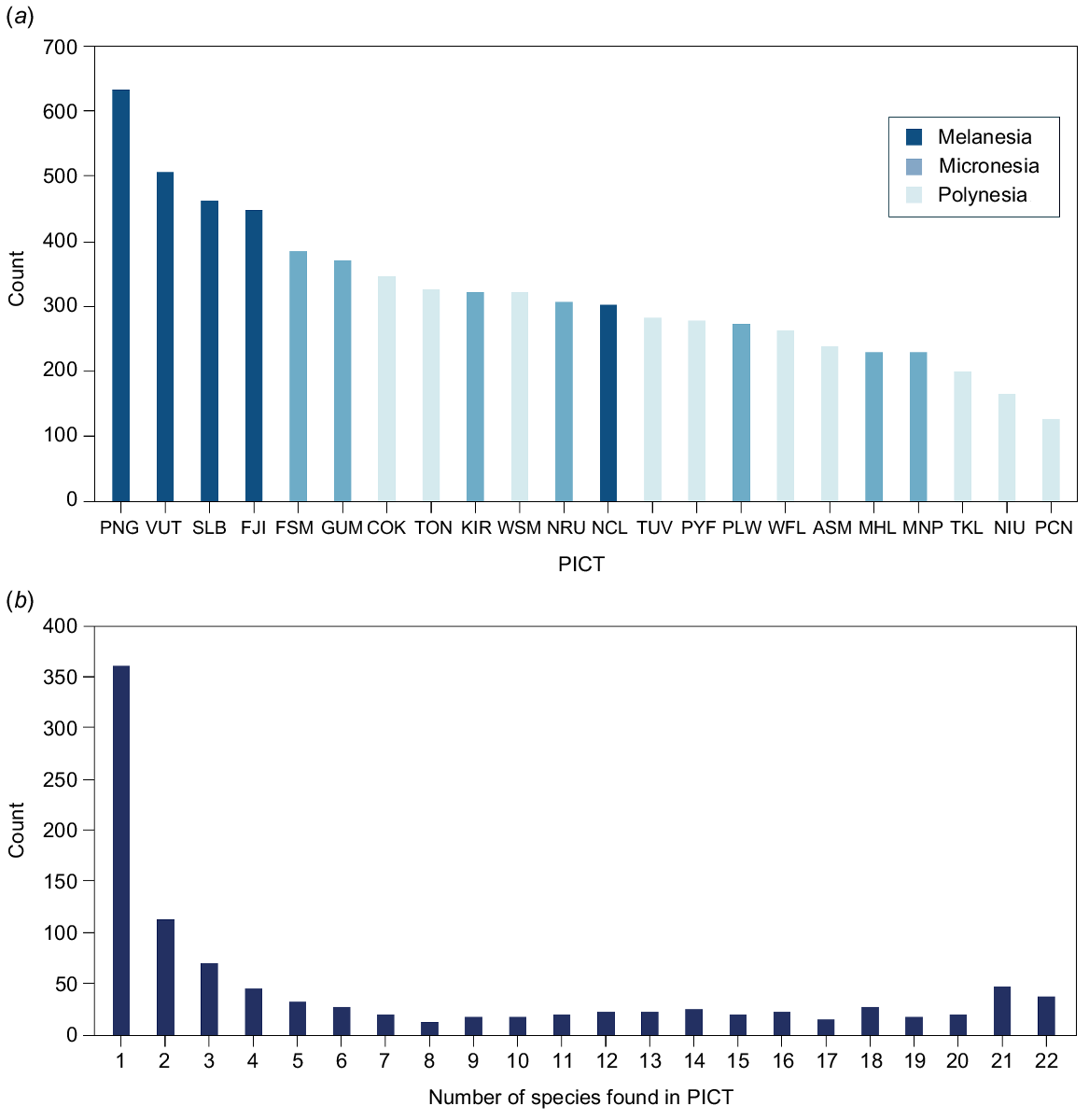
Broadly, the number of foodfish species in each PICT followed subregional patterns reported in the broader species pool (Fig. 3b), with those in the larger and more western islands of Melanesia having the most species and Polynesian PICTs the fewest species (Fig. 4a). The most species were reported from PNG (633 species), in the heart of the Coral Triangle (Veron et al. 2011) and the fewest (129) from Pitcairn Islands on the region’s subtropical eastern margin. Although a small number of species (39) was recorded in all PICTs, most were reported from a subset of PICTs (Fig. 4b). If subtropical Pitcairn Islands are excluded, a further 22 species were recorded in all the remaining PICTs. Approximately half (53%) of all species were reported from three or fewer PICTs, while over 35% were reported in only one PICT (Fig. 4b).
Foodfish occurrence by PICT and species: (a) number of species reported as foodfish in each of 22 PICTs and (b) number of species reported as caught in 1 to 22 PICTs. For example, 370 species were reported from one PICT only but not the same PICT. PICT abbreviations are as per Fig. 2.
The scientific names of 148 (14%) of the 1031 species in the Pacific foodfish list have changed since the original surveys were done. Some species (e.g. Atropus hedlandensis, the Bumpnose Trevally) have had several name changes, resulting in a total of 212 changes being recorded. Significant recent taxonomic reworkings include Kimura et al.’s (2022) revision of the genus Carangoides and that of the goatfish family Mullidae by Uiblein et al. (2024). Given that the surveys were implemented over many years but mostly in the last 20 years (Table S2), this percentage of change cannot be interpreted simply as a rate of change. Nevertheless, the dynamic nature of taxonomy as a discipline is highlighted.
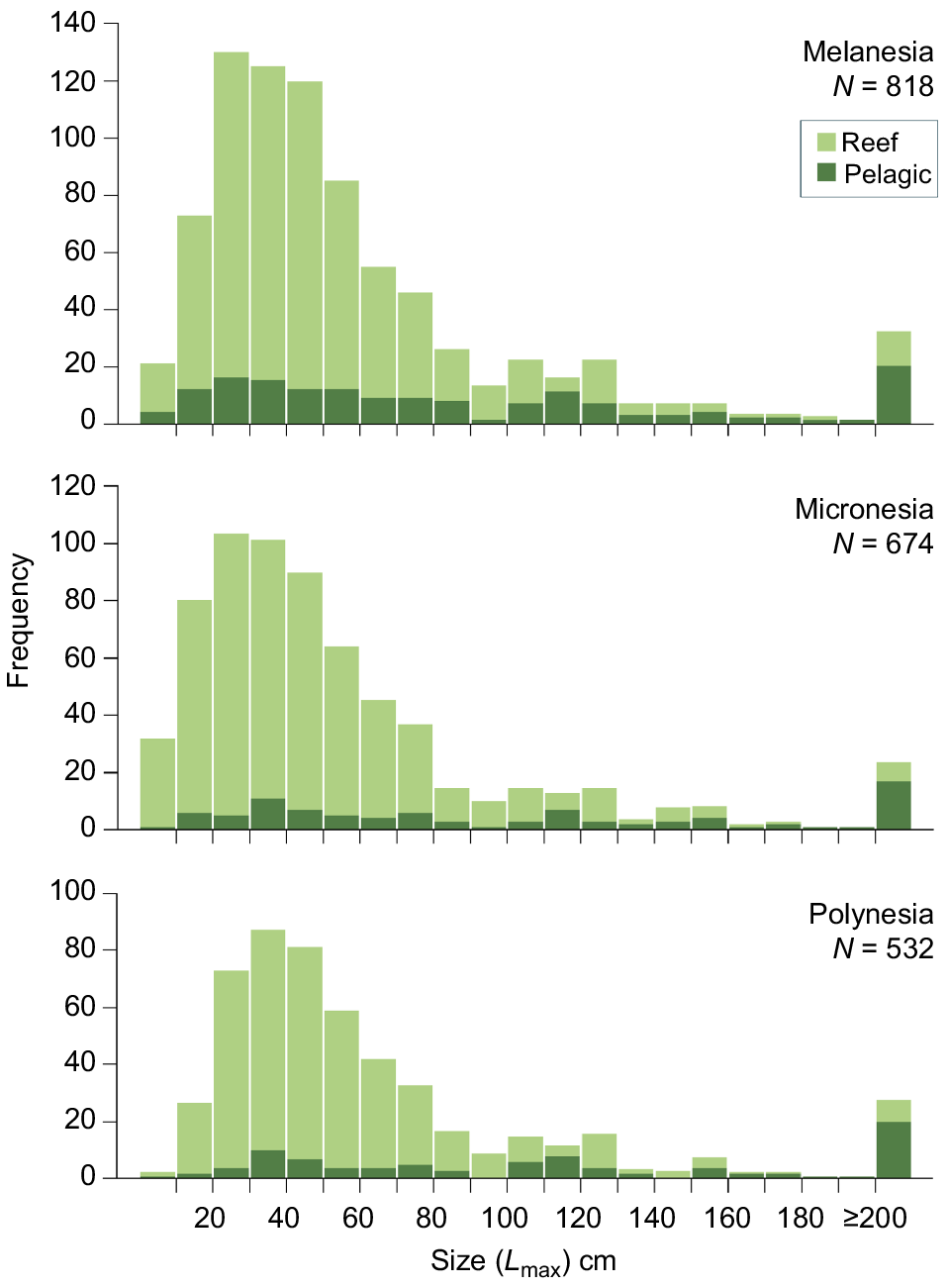
The maximum sizes of fish species reported among the three subregions were broadly similar (Fig. 5). Maximum sizes were right skewed for reef fish in all subregions, and nearly 50% of foodfish had maximum sizes between 20 and 50 cm. These were most skewed in Micronesia where 48% of reef fish had Lmax sizes of <40 cm compared to Melanesia (44%) and Polynesia (38%). Comparatively, the Lmax for pelagic species were more evenly distributed and in all subregions the highest counts of pelagic species recorded as foodfish were >200 cm. This included several tuna species, and scombrids and marlins, among others.
English common names and eponyms
There was near complete alignment of English common names among the Pacific foodfish list, FAO, FishBase, SPC and Moore and Colas (2016) (see Pacific foodfish list). English common names for just 39 species differed among some subset of these lists (Table S3). Of these, alternative names were used to avoid duplication in five species (e.g. Silver Sweeper to Blackspot Sweeper for Pempheris oualensis) and two were changed because of taxonomic revisions that differentiated previously widely distributed species (e.g. the previously circumglobal species Lampris guttatus (Opah) was split into five species, of which only the Bigeye Pacific Opah (Lampris megalopsis) was within scope for the Pacific foodfish list; Underkoffler et al. 2018). These changes, along with 42 minor changes to the form of the common name (Page et al. 2023) were made in FishBase (Table S4).
The source lists contained different subsets of species and common names were assigned according to the protocol described above. In total, the English common names of 654 species (63%) in the Pacific foodfish list were taken from the FAO list. A further 18 were drawn from AmFS, and 57 from Moore and Colas (2016) and SPC. Names in AmFS differed for 28 species but the FAO name was used in all but two cases: the Opah, as described above, and the Whitecheek Surgeonfish (Acanthurus nigricans) where the English common name from SPC and Moore and Colas (2016) was used (Table S3). The remainder (293 species) were taken from FishBase and therefore the literature but principally the sources listed above.
A total of 145 species (including monotypic genera), seven genera and one family represented in the Pacific foodfish had scientific names honoring a person, in most cases an ichthyologist or naturalist (Table S5). A further four species had eponymous common names but not scientific names. In addition, six species were named after mythological figures, notably Ctenogobiops tangaroai, named after Tangaroa, the Polynesian god of the sea. Two species were named after institutions: the Bicolored Foxface (Siganus uspi) was named after the University of the South Pacific and the Roughear Scad (Decapterus tabl) was named after the Tropical Atlantic Biological Laboratory in the USA. Only 44 of 145 eponymous species names were reflected in the English common name and four species had eponymous English common names but not scientific names (Table S5), three of which were named after the person that described the species. The fourth, the brightly patterned Halfmoon Picassofish (Rhinecanthus lunula) was presumably named after Pablo Picasso.
There was a clear disjunction at the turn of the 19th and 20th centuries in the nationality of the people honored by species names. Slightly more than half (61%) of the eponyms in the foodfish list were named in the 18th and 19th centuries and all after Europeans (excluding mythical figures; Table S5). In contrast, Europeans accounted for only 20% of species names in the 20th and current centuries – most were named after people from Pacific Rim countries, particularly USA, Australia and Japan. The first eponym used in the Pacific foodfish list was, fittingly, coined by Carl Linnaeus in 1766 and the last, in 2021, was named after Brian Bowen, an ichthyologist from Hawai‘i.
Overall, citizens of European countries accounted for 65% of eponyms in the foodfish list, followed by the USA (21%) and Australia (6%). Notable among those with species named after them are Mr Jean Tapu, a world champion spearfisher from the Tuamotu archipelago, and possibly the only Pacific Islander in the list; and three women, Mary Louisa Putnam, a patron of the Academy of Natural Sciences in Davenport, Iowa (in 1905), Joyce K. Allan, an Australian conchologist and scientific illustrator (1931), and Katherine A. Meyer, an American citizen scientist (1979). Interestingly, Odontanthias tapui, named after Mr Tapu, is one of only three species with eponymous scientific names to not have an English common name.
Representativeness of the Pacific foodfish list
Within the same inclusion criteria used to compile the foodfish list, a total of 3847 species of fish from 207 families is reported to occur in the Pacific region (see ‘Pacific biodiversity list’ available at https://pacificdata.org/). The highest diversity of fish was found in Melanesia (3392 species), followed by Micronesia (2198) and Polynesia (1930; Fig. 3b), and 2816 species from 175 families of these did not appear in national surveys of catches.
Families that included larger, diurnal fish species appeared more frequently than smaller, nocturnal and cryptic fishes in the foodfish list. Many taxa of smaller fishes were poorly represented in catch and market surveys, most notably gobies, blennies and triplefins (Fig. 6a). Many of these smaller fishes are caught as food by gleaning and children but these types of fishing events are often under-reported in national surveys, despite their importance to food provisioning. Nocturnal and cryptic taxa, including some species of larger fish such as moray and conger eels, and those not targeted by fishers, such as pipefish and seahorses are also under-represented. This trend is also represented within the group of selected families, such as scorpionfishes and anchovies (Fig. 6b), and within families excluded from the plot because no species were reported in the foodfish list, such as dragonets and jawfish. At the other end of the spectrum, species of larger fish such as lutjanids, carangids, lethrinids and sphyraenids were well represented in counts of species and the great majority species in these families were recorded in catches (Fig. 6b, c). All 11 species of tuna reported from the region were recorded in catches.
Number of species recorded in foodfish surveys as a subset of total species counts in: (a) the 10 families with the lowest proportion of total species represented in the Pacific foodfish list (ordered by descending total species counts), (b) 15 selected families, and (c) the 10 families/tribes with the highest proportion of total species represented in the Pacific foodfish list. Numbers within broken bars indicate total number of Pacific species in the family. Summary excludes families with fewer than 10 species and those that do not appear in the foodfish list.

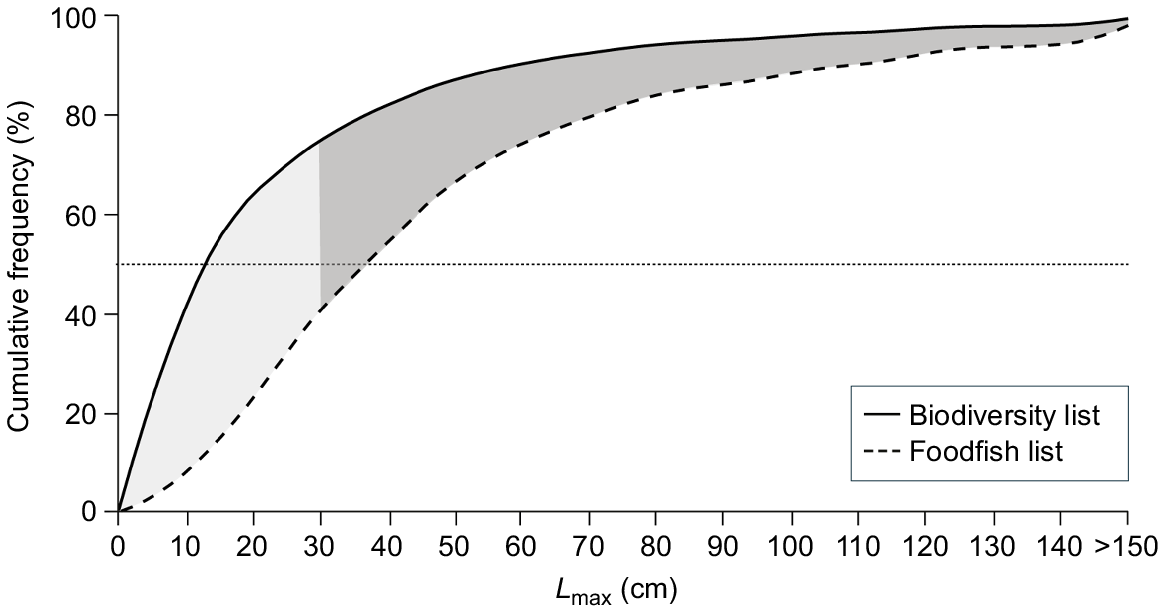
Irrespective of identity, relatively fewer species with a maximum recorded length of <20 cm were reported in catch and market surveys despite these small fish dominating species counts (Fig. 7). Approximately 55% of total species across the region had a Lmax of <20 cm, yet less than 20% of species with a Lmax of <20 cm were recorded in the surveys. In contrast, half of the species with Lmax ≥ 30 cm were recorded as foodfish (Fig. 7).
Frequency histograms of maximum sizes (Lmax) of species occurring in the region (dark bars) compared to the subset caught as foodfish (light bars).
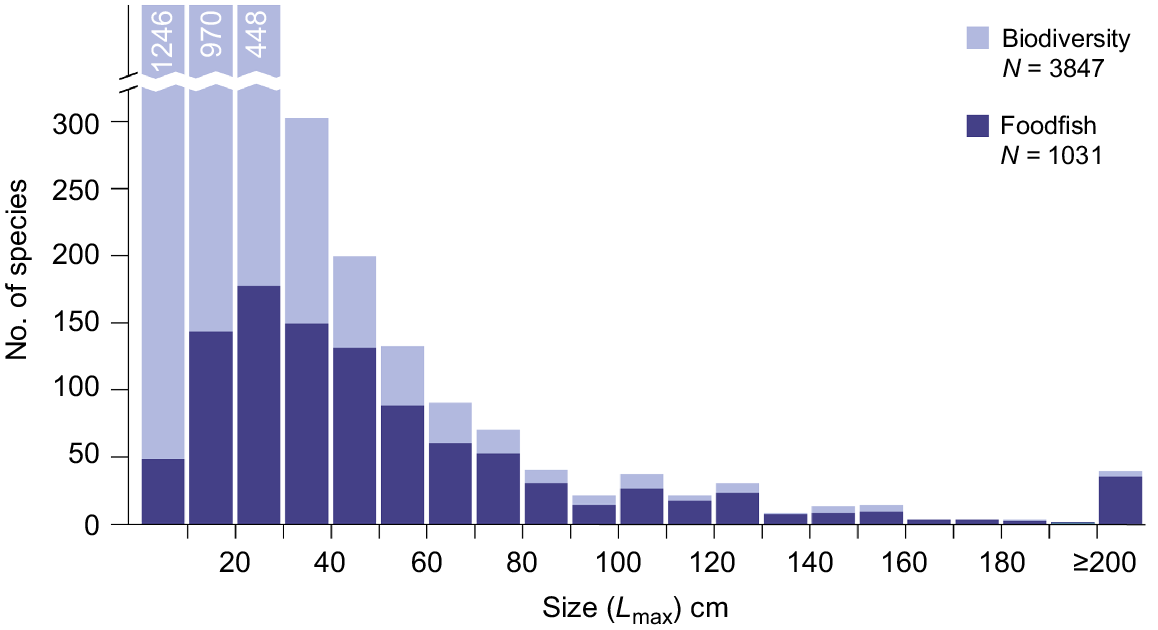
Considering the diversity of fishes in the region, disproportionately more larger species are caught for food than are present in the region – 57% of all fish species in the region have a maximum recorded size <20 cm, while only 19% of foodfish species are less than 20 cm at Lmax (Fig. 8). Half of all fish in the region are smaller than 15 cm at Lmax while half of all foodfish are smaller than 40 cm (Fig. 8). If all fish species >20 cm at Lmax and not currently in the foodfish list were added, there would be 1666 species in the list; the comparable estimates for 30 and 50 cm are 1406 and 1184 (Fig. 8). The deficit between the current foodfish list and the potential total is interpreted as a mixture of differences in catchability of species, deficits in sampling regimes and differences in cultural preferences for types of fish among PICTs.
Discussion
We provide a comprehensive, updated list of standardized names of Pacific marine foodfish. The list affirms the widely recognized diversity of foodfishes in the region and provides a foundation for improved estimates of the types and quantities of fish caught. An agreed set of English common names for the 1031 species in the foodfish list will, for example, enable more effective monitoring of fish catches as technology in this area moves towards centralized artificial intelligence-driven platforms (Shedrawi et al. 2024). Such platforms operate at a regional scale and therefore agreed scientific and English common names facilitate the aggregation of data across PICTs.
We speculate that, from the current count of 1031 species, the list will continue to expand as sampling becomes more extensive and more species are recorded, but will likely asymptote toward the total number of available species of larger fishes unless creel surveys target gleaning fisheries, and therefore the many small species occurring in the region. National lists reflect the relative abundance of species, their catchability and the nature of sampling. Overall, there appears to be a bias toward larger fish that are sold rather than caught for home consumption. Across the whole region, self-caught fish account for nearly half the fish acquired by households (Sharp and Andrew 2024). As more community fisheries are surveyed, including harvesting by gleaning, more smaller fishes will be included. Although the current coverage of species with an Lmax < 20 cm may accurately represent catches of species, this accounts for a relatively small proportion of the small fish species present. Compiling lists for freshwater fishes, sharks and rays, invertebrates and other aquatic foods remains a significant challenge before the aquatic foods of the Pacific region can be described in a common, systematic way.
The list will also enable a range of future research. Distinct derivative work will be to quantify the relative importance of foodfish species: we speculate that fewer than 100 species will account for more than 90% of catch and that the species that dominate catches will vary enormously at a range of scales. At subnational scales, islands and atolls with lagoons and/or extensive mangroves will have a different suite of species than those with only small fringing reefs (Smallhorn-West et al. 2022; Campbell et al. 2024). We predict that analogous patterns will be found among countries with contrasting reef types (comparing, for example, Niue and Tonga), and that these patterns will be overlayed with broader biogeographic patterns in the relative importance of species.
The foodfish list is conservative for several reasons. The list underestimates the diversity observed in surveys because some species are difficult to identify in the field or from photographs and were therefore recorded as ‘sp.’ (e.g. species of Acanthurus, Gerres and Myripristis). A proportion of the species excluded because these were not recorded from the region will have been misidentified and therefore may have been species not recorded in the current version of the list. Furthermore, although a wide range of fishing methods was used, species that are small, day-time cryptic, nocturnal, pelagic and those occurring on mesophotic and rariphotic reefs will be underestimated by both fishery-dependent surveys and the broader biodiversity/ecological surveys of fish (Ackerman and Bellwood 2000; Depczynski and Bellwood 2003; Stefanoudis et al. 2019). As a further source of conservatism, some of the euryhaline species excluded may have been included as ‘marine’ by other authors for other purposes (e.g. mullets in genus Cestraeus).
The creation of the Pacific foodfish list is not completely divorced from the ongoing international debate about the appropriateness of scientific and English common names of animals and plants. Strong views have been expressed for and against the revision of names deemed to be inappropriate (Smith and Figueiredo 2022; Tracy 2022; Cheng et al. 2023). Although offering opinions about the appropriateness of names is beyond the scope of this article, some points may be made that are relevant to such considerations.
The 145 eponymous species names recorded represent 14% of the total species count, which is slightly less than the global percentage of 20% (Ceríaco et al. 2023). Approximately two-thirds of these scientific names are more than 100 years old and only 17 have been named in the past 50 years. These names therefore must be viewed within the historical context in which they were created – mostly by citizens of colonizing European powers or, more recently, by American and Australian taxonomists. Furthermore, the norms of taxonomy are guided by a set of principles and rules articulated in the International Code of Zoological Nomenclature. A central aim of the Code is to provide universality and continuity to scientific names of animals. The Commission governing the Code has clarified that making judgements about the ethical worth of proposed scientific names is beyond their mandate (Ceríaco et al. 2023). Although the debate continues, for practical purposes the status quo is unlikely to change, and the retrospective changing of scientific names of Pacific foodfish in the foreseeable future is therefore unlikely.
Only 43 (4%) species in the foodfish list have eponymous English common names. Most of these species are named for ichthyologists, naturalists or others involved in the scientific process. Beyond eponyms, the Pacific foodfish list contains five English common names that may be perceived as offensive: Chinamanfish (Symphorus nematophorus), Moorish Idol (Zanclus cornutus), Quakerfish (Malacanthus brevirostris), Hawaiian Ladyfish (Elops hawaiensis) and Redcoat (Sargocentron rubrum). All five names are used in FAO’s list of common names. To paraphrase Winker (2022), in considering the appropriateness of English common names, a balance needs to be struck between the advantages of stability and the desire to improve names by changing them.
We note that the current list of 1031 species provides an incomplete basis for analysing the English common names of fishes in the region. Ongoing work extends the current summary to the wider pool of 3847 Pacific fish species that could potentially contribute as foodfish and offers perspectives on the changing role of English common names for fish in the region with particular reference to lists of national standard English names (N. L. Andrew, et al., unpubl. data).
In some respects, forming opinions about the suitability or otherwise of English common names for Pacific foodfish is a red herring because such names are little used in everyday speech in the region. The oddness of many names is exemplified by reference to northern hemisphere mammals (e.g. hind, squirrelfish, goatfish and ponyfish) and by European cultural references (e.g. cardinal, emperor and fusilier). The primary purpose of English common names in the Pacific foodfish list is to serve as an identifier for less approachable scientific names and improve communication about species among PICTs. The great majority of these names are descriptive of the fish but they need not be so. In some PICTs, e.g. Vanuatu, English common names or FAO three letter species codes are used by enumerators in creel surveys rather than scientific names. Although English is an official language in 19 of the 22 PICTs, and many people are fluent, Pacific names, both in national languages and the hundreds of local languages and dialects in the region have primacy in everyday life.
Conclusion
The Pacific foodfish list of 1031 species affirms the widely recognized diversity of foodfish in the region and provides a foundation for more comprehensive estimates of the types and quantities of fish caught. The English common names assigned to species show a very high degree of concordance with international lists and therefore contribute both to the widely held ambition to establish universal common names for fish and fostering improved communication about fish used for food within the region. At the time of writing, the list is under consideration by national institutions as a standard list for the region.
Data availability
The Pacific foodfish list and related tables are available from the SPC Pacific Data Hub (https://pacificdata.org/). Search for ‘Pacific foodfish list’. The list will be updated from time to time as new species are reported and errors corrected.
Declaration of funding
This work was funded by the Australian Government through ACIAR project FIS/2020/172 and the CGIAR Research Initiative on Aquatic Foods funded by CGIAR Trust Fund donors.
Acknowledgements
In large part this article is based on the hard work of others. We acknowledge and thank the innumerable fishers and retailers who allowed their fish to be sampled, and the fisheries officers and enumerators who recorded that information over decades of work. The summaries were enabled by, among others, three databases: Eschmeyer’s Catalog of Fishes, FishBase and ETYFish. We acknowledge the people who built and curate these public resources. We are grateful to SPC and national agencies for providing access to unpublished creel and market survey data. We thank the following for sharing data and/or expertise: Tim Adams (SPC), Justin Aiafi (WSM), Mike Batty (TUV), Yannick Chancerelle (PYF), Javier Cuetos-Bueno (FSM, MHL, PLW), Lorel Dandava-Oli (PNG), Donald David (SPC), Gerry Davis (NOAA), Aurelie Delisle (UOW), Bruno Deprez (SPC), David Fatai (SLB), Faasulu Fepuleai (WSM), Kirby Gonzalo Morejohn (COK), Lesley Gould (TKL), Doug Hoese (AUS), Geoff Jones (JCU), Watisoni Lalavanua (SPC), Filipo Makolo (TUV), Christina Muller Karanassos (PLW), Pita Neihapi (VUT), Lina Pandihau (PNG), Christian Ramofafia (SLB), Gilles Siu (PYF), Andrew Smith (SPC) and Brent Tibbatts (GUM). All errors remain ours.
References
Ackerman JL, Bellwood DR (2000) Reef fish assemblages: a re-evaluation using enclosed rotenone stations. Marine Ecology Progress Series 206, 227-237.
| Crossref | Google Scholar |
Andrew N, Campbell B, Delisle A, Li O, Neihapi P, Nikiari B, Sami A, Steenbergen D, Uriam T (2020) Developing participatory monitoring of community fisheries in Kiribati and Vanuatu. SPC Fisheries Newsletter 162, 32-38.
| Google Scholar |
Bell JD, Kronen M, Vunisea A, Nash WJ, Keeble G, Demmke A, Pontifex S, Andréfouët S (2009) Planning the use of fish for food security in the Pacific. Marine Policy 33, 64-76.
| Crossref | Google Scholar |
Bell JD, Allain V, Allison EH, Andréfouët S, Andrew NL, Batty MJ, Blanc M, Dambacher JM, Hampton J, Hanich Q, Harley S, Lorrain A, McCoy M, McTurk N, Nicol S, Pilling G, Point D, Sharp MK, Vivili P, Williams P (2015) Diversifying the use of tuna to improve food security and public health in Pacific Island countries and territories. Marine Policy 51, 584-591.
| Crossref | Google Scholar |
Bell JD, Albert J, Amos G, Arthur C, Blanc M, Bromhead D, Heron SF, Hobday AJ, Hunt A, Itano D, James PAS, Lehodey P, Liu G, Nicol S, Potemra J, Reygondeau G, Rubani J, Scutt Phillips J, Senina I, Sokimi W (2018) Operationalising access to oceanic fisheries resources by small-scale fishers to improve food security in the Pacific Islands. Marine Policy 88, 315-322.
| Crossref | Google Scholar |
Bellwood DR, Hughes TP, Connolly SR, Tanner J (2005) Environmental and geometric constraints on Indo-Pacific coral reef biodiversity. Ecology Letters 8, 643-651.
| Crossref | Google Scholar |
Binohlan C, Froese R, Pauly D, Reyes R (2011) The LENGTH-LENGTH table in FishBase (Version 04/2011). Available at https://www.fishbase.se/manual/english/PDF/FB_Book_CBinohlan_Length-Length_RF_JG.pdf [accessed 22 October 2023]
Bogard JR, Andrew NL, Farrell P, Herrero M, Sharp MK, Tutuo J (2021) A typology of food environments in the Pacific region and their relationship to diet quality in Solomon Islands. Foods 10, 2592.
| Crossref | Google Scholar |
Campbell B, Steenbergen D, Li O, Sami A, Nikiari B, Delisle A, Neihapi P, Uriam T, Andrew N (2024) Characterising a diversity of coastal community fisheries in Kiribati and Vanuatu. Fish and Fisheries 25, 837-857.
| Crossref | Google Scholar |
Ceríaco LM, Aescht E, Ahyong ST, Ballerio A, Bouchard P, Bourgoin T, Dmitriev D, Evenhuis N, Grygier MJ, Harvey MS, Kottelat M (2023) Renaming taxa on ethical grounds threatens nomenclatural stability and scientific communication: Communication from the International Commission on Zoological Nomenclature. Zoological Journal of the Linnean Society 197, 283-286.
| Crossref | Google Scholar |
Cheng SJ, Gaynor KM, Moore AC, Darragh K, Estien CO, Hammond JW, Lawrence C, Mills KL, Baiz MD, Ignace D, Khadempour L, McCary MA, Rice MM, Tumber-Dávila SJ, Smith JA (2023) Championing inclusive terminology in ecology and evolution. Trends in Ecology & Evolution 38, 381-384.
| Crossref | Google Scholar | PubMed |
Dalzell P, Preston GL (1992) Deep Reef Slope Fishery Resources of the South Pacific: a summary and analysis of the dropline fishing survey data generated by the activities of the SPC Fisheries Program between 1974 and 1988. South Pacific Commission, Inshore Fisheries Research Project Technical Document No. 2, Noumea.
Depczynski M, Bellwood DR (2003) The role of cryptobenthic reef fishes in coral reef trophodynamics. Marine Ecology Progress Series 256, 183-191.
| Crossref | Google Scholar |
D’agata S, Mouillot D, Kulbicki M, Andréfouët S, Bellwood DR, Cinner JE, Cowman PF, Kronen M, Pinca S, Vigliola L (2014) Human-mediated loss of phylogenetic and functional diversity in coral reef fishes. Current Biology 24, 555-560.
| Crossref | Google Scholar | PubMed |
Edgar GJ, Stuart-Smith RD, Willis TJ, Kininmonth S, Baker SC, Banks S, Barrett NS, Becerro MA, Bernard ATF, Berkhout J, Buxton CD, Campbell SJ, Cooper AT, Davey M, Edgar SC, Försterra G, Galván DE, Irigoyen AJ, Kushner DJ, Moura R, Parnell PE, Shears NT, Soler G, Strain EMA, Thomson RJ (2014) Global conservation outcomes depend on marine protected areas with five key features. Nature 506, 216-220.
| Crossref | Google Scholar | PubMed |
FAO (2020) FAOSTAT statistical database. Available at https://www.fao.org/statistics/en [accessed 22 October 2023]
FAO (2024) ASFIS list of species for fishery statistics purposes. Available at https://www.fao.org/fishery/en/collection/asfis/en [accessed 14 November 2024]
Fricke R, Kulbicki M, Wantiez L, Earl C (2023) Checklist of the fishes of New Caledonia, and their distribution in the Southwest Pacific Ocean (Pisces): Fishes Checklist. Version 2.0. OBIS Secretariat. Occurrence dataset. Available at https://doi.org/10.25607/fq9y4b
Fricke R, Eschmeyer WN, Van der Laan R (Eds) (2025) Eschmeyer’s catalog of fishes: genera, species and references. Available at http://researcharchive.calacademy.org/research/ichthyology/catalog/fishcatmain.asp [last accessed 2 February 2025]
Friedlander AM, Sandin SA, DeMartini EE, Sala E (2010) Spatial patterns of the structure of reef fish assemblages at a pristine atoll in the central Pacific. Marine Ecology Progress Series 410, 219-231.
| Crossref | Google Scholar |
Friedlander AM, Caselle JE, Ballesteros E, Brown EK, Turchik A, Sala E (2014) The real bounty: marine biodiversity in the Pitcairn Islands. PLoS ONE 9, e100142.
| Crossref | Google Scholar |
GBIF (2023) Global Biodiversity Information Facility Home Page. Available at https://www.gbif.org [accessed 18 December 2023]
Goetze JS, Langlois TJ, Egli DP, Harvey ES (2011) Evidence of artisanal fishing impacts and depth refuge in assemblages of Fijian reef fish. Coral Reefs 30, 507-517.
| Crossref | Google Scholar |
Hoegh-Guldberg O, Andréfouët S, Fabricius KE, Diaz-Pulido G, Lough JM, Marshall PA, Pratchett, MS (2011) Vulnerability of coral reef in the tropical Pacific to climate change. In ‘Vulnerability of tropical pacific fisheries and aquaculture to climate change’. (Eds JD Bell, JE Johnson, AJ Hobday) pp. 251–296. (Secretariat of the Pacific Community: Noumea)
Houk P, Benavente D, Fread V (2012) Characterization and evaluation of coral reefs around Yap Proper, Federated States of Micronesia. Biodiversity and Conservation 21, 2045-2059.
| Crossref | Google Scholar |
Hubert N, Dettai A, Pruvost P, Cruaud C, Kulbicki M, Myers RF, Borsa P (2017) Geography and life history traits account for the accumulation of cryptic diversity among Indo-West Pacific coral reef fishes. Marine Ecology Progress Series 583, 179-193.
| Crossref | Google Scholar |
Iwatsuki Y, Kume M, Yoshino T (2010) A new species, Acanthopagrus pacificus from the Western Pacific (Pisces, Sparidae). Bulletin of the National Museum of Nature and Science Series A 36, 115-130.
| Google Scholar |
Kimura S, Takeuchi S, Yadome T (2022) Generic revision of the species formerly belonging to the genus Carangoides and its related genera (Carangiformes: Carangidae). Ichthyological Research 69, 433-487.
| Crossref | Google Scholar |
Myers RF, Burdick DR, Mundy BC, Lindfield SJ, Tibbatts B, Donaldson TJ (2025) New and recent records of fishes from the Mariana Islands, western Pacific Ocean. Journal of the Ocean Science Foundation 42, 15-90.
| Google Scholar |
Page LM, Bemis KE, Dowling TE, Espinosa-Pérez H, Findley LT, Gilbert CR, Hartel KE, Lea RN, Mandrak NE, Neighbors MA, Schmitter-Soto JJ, Walker HJ Jr (2023) ‘Common and scientific names of fishes from the United States, Canada, and Mexico.’ 8th edn. (American Fisheries Society, Special Publication 37: Bethesda, Maryland)
Parenti P (2021) A checklist of the gobioid fishes of the world (Percomorpha: Gobiiformes). Iranian Journal of Ichthyology 8(Suppl. 1), 1-480.
| Google Scholar |
Parravicini V, Kulbicki M, Bellwood DR, Friedlander AM, Arias-Gonzalez JE, Chabanet P, Floeter SR, Myers R, Vigliola L, D’Agata S, Mouillot D (2013) Global patterns and predictors of tropical reef fish species richness. Ecography 36, 1254-1262.
| Crossref | Google Scholar |
Pinca S, Kronen M, Friedman K, Magron F, Chapman L, Tardy E, Pakoa K, Awira R, Boblin P, Lasi F (2010) Regional assessment report: profiles and results from survey work at 63 sites across 17 Pacific Island Countries and Territories. Pacific Regional Oceanic and Coastal Fisheries Development Program (PROCFish/C/CoFish) and the Pacific Community (SPC), Noumea, New Caledonia.
Randall JE (1999) Report on fish collections from the Pitcairn Islands. Atoll Research Bulletin 461, 1-36.
| Crossref | Google Scholar |
Randall JE, Williams JT, Smith DG, Kulbicki M, Tham GM, Labrosse P, Kronen M, Clua E, Mann BS (2004) Checklist of the shore and epipelagic fishes of Tonga. Atoll Research Bulletin 502, 1-35.
| Crossref | Google Scholar |
Scharpf C (2024) The ETYFish Project. Fish Name Etymology Database. Available at https://etyfish.org/aulopiformes/ [accessed 22 October 2023]
Shedrawi G, Magron F, Vigga B, Bosserelle P, Gislard S, Halford AR, Tiitii S, Fepuleai F, Molai C, Rota M, Jalam S, Fatongiatau V, Sami AP, Nikiari B, Sokach AHM, Joy LA, Li O, Steenbergen DJ, Andrew NL (2024) Leveraging deep learning and computer vision technologies to enhance management of coastal fisheries in the Pacific region. Scientific Reports 14, 20915.
| Crossref | Google Scholar |
Siu G, Bacchet P, Bernardi G, Brooks AJ, Carlot J, Causse R, Claudet J, Clua É, Delrieu-Trottin E, Espiau B, Harmelin-Vivien M, Keith P, Leccini D, Madi-Moussa R, Parravicini V, Planes S, Ponsonnet C, Randall JE, Sasal P, Taquet M, Williams JT, Galzin R (2017) Shore fishes of French Polynesia. Cybium 41, 245-278.
| Google Scholar |
Smallhorn-West P, van der Ploeg J, Boso D, Sukulu M, Leamae J, Isihanua M, Jasper M, Saeni-Oeta J, Batalofo M, Orirana G, Konamalefo A, Houma J, Eriksson H (2022) Patterns of catch and trophic signatures illustrate diverse management requirements of coastal fisheries in Solomon Islands. Ambio 51, 1504-1519.
| Crossref | Google Scholar | PubMed |
Smith GF, Figueiredo E (2022) “Rhodes-” must fall: some of the consequences of colonialism for botany and plant nomenclature. Taxon 71, 1-5.
| Crossref | Google Scholar |
Stefanoudis PV, Gress E, Pitt JM, Smith SR, Kincaid T, Rivers M, Andradi-Brown DA, Rowlands G, Woodall LC, Rogers AD (2019) Depth-dependent structuring of reef fish assemblages from the shallows to the rariphotic zone. Frontiers in Marine Science 6, 307.
| Crossref | Google Scholar |
Tracy BH (2022) What’s in a fish species name and when to change it? Fisheries 47, 337-345.
| Crossref | Google Scholar |
Uiblein F, Williams JT, Bailly N, Hoang TA, Rajan PT (2024) Four new goatfishes (Upeneus, Mullidae, Mulliformes) from the Asian Indo-Pacific with a list of valid goatfish species and remarks on goatfish diversity. Cybium 48, 135-160.
| Crossref | Google Scholar |
Underkoffler KE, Luers MA, Hyde JR, Craig MT (2018) A taxonomic review of Lampris guttatus (Brünnich 1788) (Lampridiformes; Lampridae) with descriptions of three new species. Zootaxa 4413, 551-565.
| Crossref | Google Scholar | PubMed |
van der Laan R, Eschmeyer WN, Fricke R (2014) Family-group names of recent fishes. Zootaxa Monograph 3882, 1-230.
| Crossref | Google Scholar |
Winker K (2022) A brief history of English bird names and the American Ornithologists’ Union (now American Ornithological Society). Ornithology 139, 1-12.
| Crossref | Google Scholar |
Yeager LA, Deith MCM, McPherson JM, Williams ID, Baum JK (2017) Scale dependence of environmental controls on the functional diversity of coral reef fish communities. Global Ecology and Biogeography 26, 1177-1189.
| Crossref | Google Scholar |