Norovirus and cruise ships
Jennifer H Lun A D , Kelly-Anne Ressler B E , Mark J Ferson B C F and Peter A White A GA School of Biotechnology and Biomolecular Sciences, Faculty of Science, University of New South Wales, Sydney, NSW, Australia
B Public Health Unit, South Eastern Sydney Local Health District, Randwick, NSW, Australia
C School of Public Health and Community Medicine, University of New South Wales, Kensington, NSW, Australia
D Email: j.lun@unsw.edu.au
E Email: KellyAnne.Ressler@health.nsw.gov.au
F Email: Mark.Ferson@health.nsw.gov.au
G Email: p.white@unsw.edu.au
Microbiology Australia 38(4) 187-190 https://doi.org/10.1071/MA17065
Published: 31 October 2017
Acute gastroenteritis (AGE) is one of the most common diseases of humans in both developed and developing countries. Despite the improved safety of food handling and prevention strategies, it remains one of the most significant health burdens on society1. An estimated 1.8 million AGE-associated deaths occur annually worldwide2,3, making it the leading cause of mortality among children under the age of five years. In this article, we examine recent trends in emerging noroviruses, the ability of the virus to cause outbreaks on cruise ships and discuss the factors that influence its on-board spread.
The main agents responsible for acute gastroenteritis include viruses, bacteria, parasites and chemical agents. Across the globe, norovirus is estimated to cause 677 million gastroenteritis cases and 212 000 deaths per year4. In Australia, norovirus is now the leading cause of viral gastroenteritis following the introduction of rotavirus vaccine to the National Immunisation Program in 2006 and infects around 2.2 million people each year5. Illness is generally self-limiting, but can be severe in vulnerable populations; including children, elderly and immunocompromised individuals6,7. The main transmission routes are person to person, through the faecal-oral route, or consumption of contaminated food and water. Norovirus is highly infectious compared to other viruses, with 100–1000 virions sufficient to infect a healthy individual8. In addition, norovirus is resistant to freezing, heating and can tolerate commonly used disinfectants, including alcohol and quaternary ammonium compounds better than other viruses9,10. The combination of these properties allows norovirus to survive in external environments for long periods. The virus often attacks in closed or semi-closed environments such as cruise ships, nursing homes, childcare centres and hospitals11,12.
Norovirus was first identified in an elementary school outbreak in Norwalk, Ohio in 196813, although it took four more years before Albert Kapikian finally discovered the virus in the faecal matter of infected individuals stored from the Norwalk outbreak14. Norovirus is a non-enveloped 27–35 nm icosahedral virion, encapsulating a single stranded RNA genome of 7.5 kilobase pairs. Its genome contains three open reading frames (ORF1-3), encoding seven non-structural proteins and two capsid proteins. Based on the full-length capsid amino acid sequences, norovirus can be categorised into seven genogroups (GI–GVI) and further divided into more than 40 genotypes15. Only GI, GII and GIV have been shown to cause disease in humans, with GII the most common (90% of cases), followed by GI (5–10%), whilst GIV is rare. Although many genotypes have now been identified, one important one known as GII, genotype 4 (GII.4), is the cause of ~70% of all norovirus infections and has been responsible for all six pandemics of norovirus–associated gastroenteritis16.
Similar to influenza A, the evolution of norovirus is attributed to two main forces; antigenic drift and antigenic shift. Antigenic drift results in amino acid divergence in the protruding (P) domain of the viral capsid, allowing escape from herd immunity acquired through previous norovirus infections. Antigenic shift or recombination, is also implicated in the emergence of new viruses and is achieved through the exchange of genetic material between strains when a co-infection occurs. The common location for norovirus recombination is at the ORF1/ORF2 overlapping region, spanning the structural and non-structural region17. Both of these mechanisms drive the evolution of GII.4 viruses and have been implicated in the emergence of the GII.4 pandemic variants over the past two decades16. The first pandemic spread of a GII.4 variant was recognised in mid-1990s18, namely the GII.4 US 95/96 variant. This variant dominated for several years until a new GII.4 strain (Farmington Hills 2002) emerged and became predominant19. Since then, four new GII.4 pandemics variants have emerged around every 2–3 years and include; Hunter 200420, Den Haag 2006b21, New Orleans 200922 and Sydney 201223,24. Previous studies have shown GII.4 strain is the most commonly identified genotype on cruise ships25.
Norovirus is notorious for causing outbreaks on cruise ships all over the world, affecting hundreds of passengers and leads to considerable economic costs for the industry26–30. The cruise ship industry has an important economic benefit to Australia, bringing in thousands of passengers each year from their hinterlands to port cities, resulting in an estimated tourist income of $1.74 billion31. Since 2007, the number of cruise ship arrivals into Sydney has trebled and their destinations abroad have doubled, with an average of 200 cruise ships berthing in Sydney each year, peaking at 320 in 2016 (Table 1). Despite the strict sanitation measures on ships, around 5.3% (yearly range 3.1%–9.0%) of ship arriving in Sydney have reported outbreaks of gastroenteritis (unpublished data, Public Health Unit, SESLHD), with norovirus commonly identified as the main culprit (Table 1). A recent investigation in the United State, from 2008 to 2014, showed norovirus is responsible for 97% (92/95) of acute gastroenteritis on cruise ships25. In fact, cruise ships have been touted as sentinel surveillance settings for new norovirus strains, with pandemics ensuing soon after the number of cruise ship outbreaks increase.
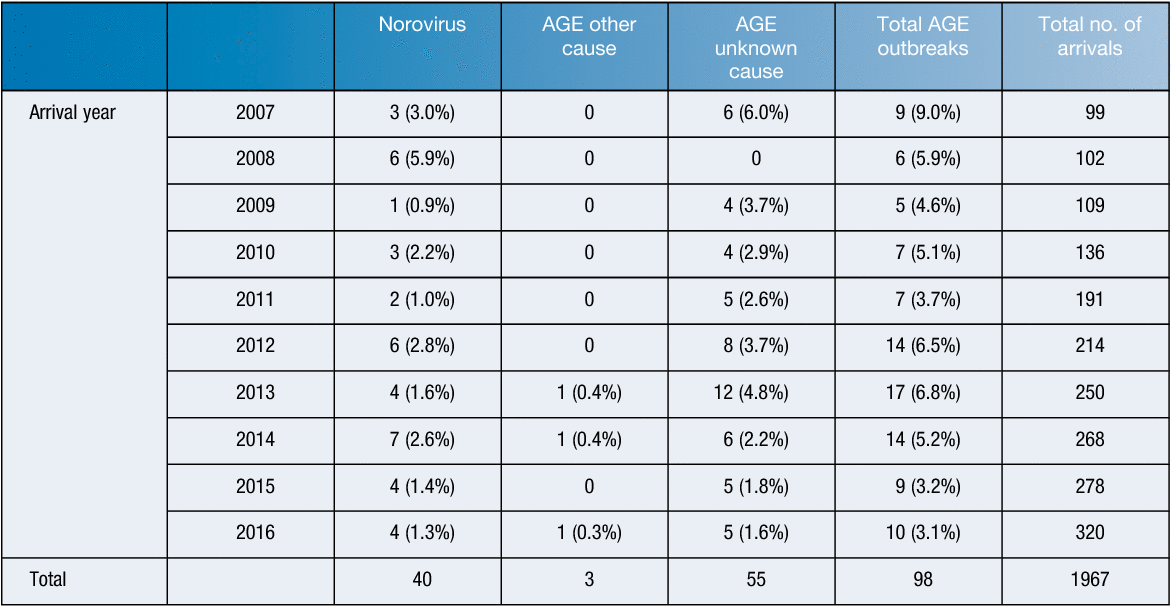
|
In attempts to prevent norovirus outbreaks on cruise ships and help eliminate the risks person-to-person spread associated with buffet style meals, some cruise ship companies have rolled out dine-in restaurants with designated wait-staff. Hand washing and the use of hand sanitisers is another strategy for outbreak prevention on ships32,33. In Australia, on-board gastroenteritis outbreaks exceeding a threshold of 3% of the ship’s passengers and crews affected are required under the Biosecurity Act 2015 (Commonwealth) to be reported to the Australian Department of Agriculture and Water Resources (DAWR). Following this notification, DAWR Biosecurity Officers inform the state or territory health authority, with subsequent implementation of prevention and control protocols. Among other things, these include: cleaning of infected cabins and more frequent laundering of linen; increased cleaning and disinfection in common areas, including the cruise terminal; deployment of external public health sanitation squads and delayed boarding of new passengers to allow additional sanitation activities. Two major classes of disinfectant are used on board cruise ships to ensure inactivation of virions, hydrogen peroxide and chlorine based sanitisers, and they can be used for decontamination of both non-food contact surfaces and food-contact surfaces.
In 2016, three new recombinant viruses emerged in Australia: GII.P4 New Orleans 2009/GII.4 Sydney 2012, GII.P16/GII.4 Sydney 2012 and GII.P16/GII.2. Due to the lack of herd immunity against these novel hybrid viruses, an increase in gastroenteritis outbreaks was observed in the population in the winter period. Over the past six years, 12 cruise ship outbreaks were investigated further to identify the aetiological agent. The GII.4 Sydney pandemic norovirus was responsible for five (42%), and the newly identified recombinant GII.P16/GII4 Sydney 2012 caused two (17%) (Table 2). In recent times, the impact of pandemic Sydney 2012 variant has declined, likely due to increased herd immunity, but viruses with the capsid GII.4 Sydney have continued to thrive in the semi-closed cruise ship environment and remain a scourge to both passengers and the cruise ship industry alike.
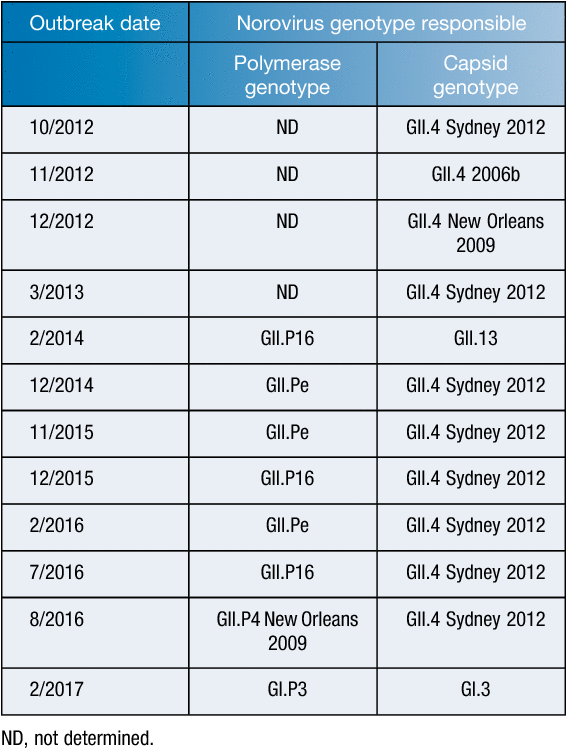
|
Cruise ships are also commonly susceptible to subsequent outbreaks once an initial outbreak has struck. A study in Europe of 13 ships over a 28-week period showed each ship had between 1 and 12 outbreaks (mean = 3.46)34. In the case of cruise ship outbreaks, great debate surrounds whether the norovirus outbreak was sourced through embarking passengers, or if the virus was already on board from the previous outbreak. After the first infection is introduced on to a ship, an outbreak is likely to ensue through person-to-person transmission. With the regular changing of passengers, noroviruses on board that avoid disinfection could repeatedly infect a new susceptible population and even exhaustive control measures might not be enough to avoid this. A number of risk factors for repeated outbreaks have been identified: these include possible contact between boarding and disembarking passenger groups, inappropriate cleaning for norovirus elimination35, and passengers refusing to be isolated to prevent further transmission of the virus36.
In conclusion, norovirus is a common cause of gastroenteritis on cruise ships due to the closed setting, its low infectious dose and its ability to survive in the environment. Norovirus evolves through antigenic drift and shift to create new strains capable of escaping herd immunity, resulting in increases in outbreaks of AGE around the globe. These emerging viruses often appear early on cruise ships34,37, therefore active surveillance is vital to protect ship’s passengers and prevent outbreaks.
Acknowledgements
We thank all the cruise ships for their cooperation and provision of samples. We also thank the NSW Public Health department and Juan Merif (Department of Microbiology, Prince of Wales Hospital, Sydney, Australia) for organising the samples to be genotyped at UNSW, and for providing relevant outbreak information. This work was supported through National Health and Medical Research Council project grants (APP1083139 and APP1123135). Jennifer Lun was supported by an Australian Postgraduate Award and a Water Research Australia scholarship.
References
[1] Patel, M.M. et al. (2008) Systematic literature review of role of noroviruses in sporadic gastroenteritis. Emerg. Infect. Dis. 14, 1224.| Systematic literature review of role of noroviruses in sporadic gastroenteritis.Crossref | GoogleScholarGoogle Scholar |
[2] Bryce, J. et al. (2005) WHO estimates of the causes of death in children. Lancet 365, 1147–1152.
| WHO estimates of the causes of death in children.Crossref | GoogleScholarGoogle Scholar |
[3] Pieścik‐Lech, M. et al. (2013) Review article: the management of acute gastroenteritis in children. Aliment. Pharmacol. Ther. 37, 289–303.
| Review article: the management of acute gastroenteritis in children.Crossref | GoogleScholarGoogle Scholar |
[4] Pires, S.M. et al. (2015) Aetiology-specific estimates of the global and regional incidence and mortality of diarrhoeal diseases commonly transmitted through food. PLoS One 10, e0142927.
| Aetiology-specific estimates of the global and regional incidence and mortality of diarrhoeal diseases commonly transmitted through food.Crossref | GoogleScholarGoogle Scholar |
[5] Gibney, K.B. et al. (2014) Disease burden of selected gastrointestinal pathogens in Australia, 2010. Int. J. Infect. Dis. 28, 176–185.
| Disease burden of selected gastrointestinal pathogens in Australia, 2010.Crossref | GoogleScholarGoogle Scholar |
[6] Hutson, A.M. et al. (2004) Norovirus disease: changing epidemiology and host susceptibility factors. Trends Microbiol. 12, 279–287.
| Norovirus disease: changing epidemiology and host susceptibility factors.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD2cXksVeltrs%3D&md5=2ce95d7077e0ec3f7e8074d21bea5881CAS |
[7] Payne, D.C. et al. (2013) Norovirus and medically attended gastroenteritis in US children. N. Engl. J. Med. 368, 1121–1130.
| Norovirus and medically attended gastroenteritis in US children.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3sXksVGkurk%3D&md5=928c6fd2d8431085b5a3630faffd9fd2CAS |
[8] Phan, T.G.T.S. et al. (2006) Detection and genetic characterization of norovirus strains circulating among infants and children with acute gastroenteritis in Japan during 2004–2005. Clin. Lab. 52, 519–525.
| 1:CAS:528:DC%2BD28Xht1Wjt7jL&md5=7eb4889bd2f7ae1e9263b72e5598d738CAS |
[9] Donaldson, E.F. et al. (2008) Norovirus pathogenesis: mechanisms of persistence and immune evasion in human populations. Immunol. Rev. 225, 190–211.
| Norovirus pathogenesis: mechanisms of persistence and immune evasion in human populations.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD1cXht1CmtbnN&md5=cf690eec6264597afd7610a60b4d8b5eCAS |
[10] Siebenga, J.J. et al. (2007) Epochal evolution of GGII.4 norovirus capsid proteins from 1995 to 2006. J. Virol. 81, 9932–9941.
| Epochal evolution of GGII.4 norovirus capsid proteins from 1995 to 2006.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD2sXhtVeht7fN&md5=3b00298cd935734fa38f41ccc2bc5865CAS |
[11] Harris, J.P. et al. (2010) Infection control measures for norovirus: a systematic review of outbreaks in semi-enclosed settings. J. Hosp. Infect. 74, 1–9.
| Infection control measures for norovirus: a systematic review of outbreaks in semi-enclosed settings.Crossref | GoogleScholarGoogle Scholar | 1:STN:280:DC%2BC3c%2Fjs1yntQ%3D%3D&md5=3ef0c68dba457bb9ca3295fbfee5825bCAS |
[12] Thornton, S. (2002) Detection of Norwalk-life virus infection aboard two US Navy ships. Mil. Med. 167, 826.
[13] Adler, J.L. and Zickl, R. (1969) Winter vomiting disease. J. Infect. Dis. 119, 668–673.
| Winter vomiting disease.Crossref | GoogleScholarGoogle Scholar | 1:STN:280:DyaF1M3js12isQ%3D%3D&md5=6fae71c2d4086b2aad635eb5c0166f3bCAS |
[14] Kapikian, A.Z. et al. (1972) Visualization by immune electron microscopy of a 27-nm particle associated with acute infectious nonbacterial gastroenteritis. J. Virol. 10, 1075–1081.
| 1:STN:280:DyaE3s%2FkvVOnuw%3D%3D&md5=cb77e84779369d2e73f3c0e9855d6e02CAS |
[15] Mesquita, J.R. et al. (2010) Novel norovirus in dogs with diarrhea. Emerg. Infect. Dis. 16, 980.
| Novel norovirus in dogs with diarrhea.Crossref | GoogleScholarGoogle Scholar |
[16] Eden, J.-S. et al. (2014) The emergence and evolution of the novel epidemic norovirus GII.4 variant Sydney 2012. Virology 450–451, 106–113.
| The emergence and evolution of the novel epidemic norovirus GII.4 variant Sydney 2012.Crossref | GoogleScholarGoogle Scholar |
[17] Bull, R.A. et al. (2007) Norovirus recombination. J. Gen. Virol. 88, 3347–3359.
| Norovirus recombination.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD2sXhsVersb7E&md5=a2d3600d70473f2fbc83cb60ec45ab70CAS |
[18] Noel, J.S. et al. (1999) Identification of a distinct common strain of ‘Norwalk-like viruses’ having a global distribution. J. Infect. Dis. 179, 1334–1344.
| Identification of a distinct common strain of ‘Norwalk-like viruses’ having a global distribution.Crossref | GoogleScholarGoogle Scholar | 1:STN:280:DyaK1M3ks1egsA%3D%3D&md5=8966e3468279cf60c6552ffd704ed5dbCAS |
[19] Widdowson, M.-A. et al. (2004) Outbreaks of acute gastroenteritis on cruise ships and on land: identification of a predominant circulating strain of norovirus—United States, 2002. J. Infect. Dis. 190, 27–36.
| Outbreaks of acute gastroenteritis on cruise ships and on land: identification of a predominant circulating strain of norovirus—United States, 2002.Crossref | GoogleScholarGoogle Scholar |
[20] Bull, R.A. et al. (2006) Emergence of a new norovirus genotype II.4 variant associated with global outbreaks of gastroenteritis. J. Clin. Microbiol. 44, 327–333.
| Emergence of a new norovirus genotype II.4 variant associated with global outbreaks of gastroenteritis.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD28XitFShtLk%3D&md5=b225312c996a15cff0701d2102eaf25cCAS |
[21] Eden, J.-S. et al. (2010) Norovirus GII.4 variant 2006b caused epidemics of acute gastroenteritis in Australia during 2007 and 2008. J. Clin. Virol. 49, 265–271.
| Norovirus GII.4 variant 2006b caused epidemics of acute gastroenteritis in Australia during 2007 and 2008.Crossref | GoogleScholarGoogle Scholar |
[22] Yen, C. et al. (2011) Impact of an emergent norovirus variant in 2009 on norovirus outbreak activity in the United States. Clin. Infect. Dis. 53, 568–571.
| Impact of an emergent norovirus variant in 2009 on norovirus outbreak activity in the United States.Crossref | GoogleScholarGoogle Scholar |
[23] van Beek, J. et al. (2013) Indications for worldwide increased norovirus activity associated with emergence of a new variant of genotype II.4, late 2012. Euro Surveill. 18, 8–9.
| 1:STN:280:DC%2BC3szmtV2ntg%3D%3D&md5=3bc4bb7221cff7446b11351a709dbf59CAS |
[24] Siebenga, J.J. et al. (2009) Norovirus illness is a global problem: emergence and spread of norovirus GII.4 variants, 2001–2007. J. Infect. Dis. 200, 802–812.
| Norovirus illness is a global problem: emergence and spread of norovirus GII.4 variants, 2001–2007.Crossref | GoogleScholarGoogle Scholar |
[25] Freeland, A.L. (2016) Acute gastroenteritis on cruise ships—United States, 2008–2014. MMWR Morb. Mortal. Wkly. Rep. 65, 1–5.
| Acute gastroenteritis on cruise ships—United States, 2008–2014.Crossref | GoogleScholarGoogle Scholar |
[26] Enserink, M. (2006) Gastrointestinal virus strikes European cruise ships. Science 313, 747.
| Gastrointestinal virus strikes European cruise ships.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD28Xot1Smu7c%3D&md5=268b2d3a97f6a6cc9d227110b7c7f550CAS |
[27] Ferson, M.J. and Ressler, K.-A. (2005) Bound for Sydney town: health surveillance on international cruise vessels visiting the Port of Sydney. Med. J. Aust. 182, 391–394.
[28] Morillo, S.G. et al. (2012) Rapid detection of norovirus in naturally contaminated food: foodborne gastroenteritis outbreak on a cruise ship in Brazil, 2010. Food Environ. Virol. 4, 124–129.
| Rapid detection of norovirus in naturally contaminated food: foodborne gastroenteritis outbreak on a cruise ship in Brazil, 2010.Crossref | GoogleScholarGoogle Scholar |
[29] Vivancos, R. et al. (2010) Norovirus outbreak in a cruise ship sailing around the British Isles: investigation and multi-agency management of an international outbreak. J. Infect. 60, 478–485.
| Norovirus outbreak in a cruise ship sailing around the British Isles: investigation and multi-agency management of an international outbreak.Crossref | GoogleScholarGoogle Scholar |
[30] Wang, X. et al. (2016) An outbreak of multiple norovirus strains on a cruise ship in China, 2014. J. Appl. Microbiol. 120, 226–233.
| An outbreak of multiple norovirus strains on a cruise ship in China, 2014.Crossref | GoogleScholarGoogle Scholar | 1:STN:280:DC%2BC28zjt1Wgsw%3D%3D&md5=abc1ee95eff795f347463a40f54e7e6fCAS |
[31] Smith, S. (2016) Economic impact of the cruise industry in Australia, 2015–16. Australian Cruise Association.
[32] Duizer, E. and Koopmans, M. (2005) Efficacy of ethanol-based hand rubs. J. Hosp. Infect. 61, 362–363.
| Efficacy of ethanol-based hand rubs.Crossref | GoogleScholarGoogle Scholar | 1:STN:280:DC%2BD2MnksFOgsQ%3D%3D&md5=8fad2123910df68748955b08df1b7d6aCAS |
[33] Kampf, G. and Ostermeyer, C. (2004) Efficacy of alcohol-based gels compared with simple hand wash and hygienic hand disinfection. J. Hosp. Infect. 56, 13–15.
| Efficacy of alcohol-based gels compared with simple hand wash and hygienic hand disinfection.Crossref | GoogleScholarGoogle Scholar |
[34] Verhoef, L. et al. (2008) Emergence of new norovirus variants on spring cruise ships and prediction of winter epidemics. Emerg. Infect. Dis. 14, 238.
| Emergence of new norovirus variants on spring cruise ships and prediction of winter epidemics.Crossref | GoogleScholarGoogle Scholar |
[35] Carling, P.C. et al. (2009) Cruise ship environmental hygiene and the risk of norovirus infection outbreaks: an objective assessment of 56 vessels over 3 years. Clin. Infect. Dis. 49, 1312–1317.
| Cruise ship environmental hygiene and the risk of norovirus infection outbreaks: an objective assessment of 56 vessels over 3 years.Crossref | GoogleScholarGoogle Scholar |
[36] Diskin, A.L. et al. (2014) Acute gastroenteritis and video camera surveillance: a cruise ship case report. Int. Marit. Health 65, 20–22.
| Acute gastroenteritis and video camera surveillance: a cruise ship case report.Crossref | GoogleScholarGoogle Scholar |
[37] Lopman, B. et al. (2004) Increase in viral gastroenteritis outbreaks in Europe and epidemic spread of new norovirus variant. Lancet 363, 682–688.
| Increase in viral gastroenteritis outbreaks in Europe and epidemic spread of new norovirus variant.Crossref | GoogleScholarGoogle Scholar |
Biographies
Jennifer Lun is a PhD student in the School of Biotechnology and Biomolecular Sciences at UNSW. Her PhD research has focused on understanding circulating norovirus strains and the evolutionary mechanisms that have facilitated the emergence of pandemic viruses.
Kelly-Anne Ressler is an Epidemiologist with the South Eastern Sydney Public Health Unit, holding a Bachelor of Science Nutrition and a Masters of Public Health from the University of Wollongong. Kelly has been employed by the Public Health Unit since 1998 and provides epidemiological advice to members of the infectious diseases, immunisation and environmental health teams. Since 2004, Kelly has coordinated the Cruise Ship Health Surveillance Program, which collects health surveillance data and provides response to outbreaks of infectious disease on board international vessels entering Sydney Harbour. Kelly also has an interest in the epidemiology of sexually transmitted infections, particularly syphilis in men who have sex with men, but also in the investigation and control of foodborne disease outbreaks and emerging infections.
Professor Mark Ferson is a public health physician and paediatrician. He is the Director of the Public Health Unit, South Eastern Sydney Local Health District, and the Chair of the NSW Infectious Diseases Network, which is tasked with promoting high standards, open communication and consistency of practice across the state’s Public Health Units. He is public health medicine subspecialty editor for the Internal Medicine Journal and the ASID representative on the editorial board for Communicable Diseases Intelligence. Mark’s research interests are in the epidemiology and control of infectious diseases, with a particular focus on childcare settings, childhood vaccination, gastroenteritis viruses and the exanthemata, and on public health law and history.
Peter White is a Professor in the School of Biotechnology and Biomolecular Sciences at UNSW. His research interests include molecular epidemiology, antiviral development, viral discovery and the replication of positive-sense viruses.


