A key regulatory mechanism of antimicrobial resistance in pathogenic Acinetobacter baumannii
Felise G AdamsMolecular Microbiology Lab
College of Science and Engineering
Flinders University
Adelaide, SA 5042, Australia
Tel: +61 8 8201 5379
Email: felise.adams@flinders.edu.au
Microbiology Australia 38(3) 122-126 https://doi.org/10.1071/MA17046
Published: 9 August 2017
Acinetobacter baumannii is a Gram-negative bacterial pathogen that has become a pressing global health issue in recent decades. Although virulence factors for this pathogen have been identified, details of how they are regulated are largely unknown. One widely employed regulatory mechanism that bacteria, such as A. baumannii, have adopted is through two component signal transduction systems (TCS). TCS consist of two proteins; a histidine kinase and response regulator. The histidine kinase allows the bacterium to sense alterations in the extracellular milieu, transmitting the information to the response regulator which prompts the cell to modify gene expression levels accordingly. Bacteria can encode multiple TCS, where each system can mediate specific responses to particular conditions or stressors. Identifying those conditions in which these TCS are expressed, and the genes they regulate known as their ‘regulon’, is vital for understanding how A. baumannii survives and persists within the hospital environment or the human host during infection. As we enter the post-antibiotic era, knowledge of TCS could prove to be invaluable, as they offer an alternative target for the treatment of multidrug resistant bacterial infections.
Acinetobacter baumannii is a Gram-negative opportunistic ‘superbug’ that causes a diverse range of nosocomial infections, primarily in patients whom are immunocompromised, such as those within intensive care units1. The Acinetobacter species is one of the leading causes of bacterial pneumonia within the hospital environment2,3 and have been responsible for numerous outbreaks of nosocomial infections, worldwide4. Within Australia, A. baumannii isolates are not only confined to hospitals but are also seen within community-based settings, primarily in tropical regions of Northern Australia5,6. These community-acquired infections have been shown to be predominant in individuals whom have underlying medical conditions such as diabetes mellitus or other risk factors including excessive alcohol consumption6.
High levels of innate and acquired multidrug resistance mechanisms represent a leading factor in the pathogenic success of this organism. Intrinsic resistance mechanisms include low outer membrane permeability, production of chromosomally-encoded antibiotic inactivating enzymes and efflux pump systems7,8. Acquired mechanisms contributing to resistance include the uptake of foreign resistance determinants via horizontal gene transfer9,10 and over-expression of resistance genes through introduction of insertional elements or mutations particularly within their regulators11,12. This ability of the bacterium to acquire and modulate expression of an array of antimicrobial resistance determinants provides a strong ecological advantage to survive the selective pressures typically found within the hospital setting, resulting in the growing emergence of multidrug resistant A. baumannii lineages1.
Infections caused by multidrug resistant A. baumannii isolates, particularly those resistant to carbapenems, have been linked to increased morbidity and mortality, as well as a significant rise in hospital associated costs13. Recently, the World Health Organization recognised the impending threat of carbapenem resistant A. baumannii isolates, listing them as one of the top three critical priorities for research and development towards new therapeutic treatments for antibiotic-resistant bacterial species (excluding Mycobacteria)14. Despite the risk this bacterium poses to susceptible individuals and our healthcare facilities, virulence traits including antimicrobial resistance, are not completely understood. Furthermore, the regulatory mechanisms that modulate these characteristics are even more ill-defined.
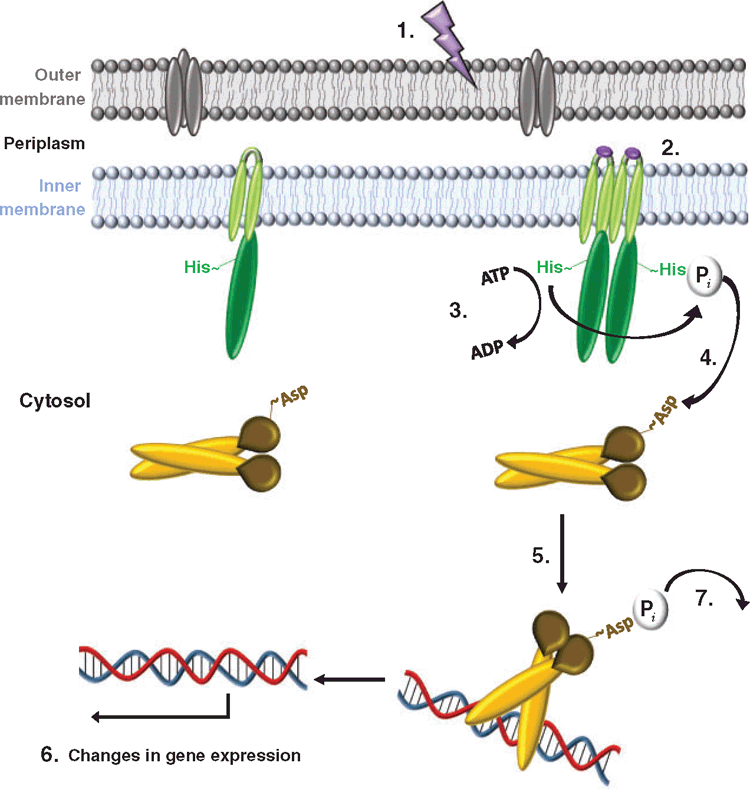
One simple yet highly sophisticated mechanism that bacteria utilise to effectively regulate the expression of virulence factors employs TCS15,16. TCS consist of two proteins; a histidine kinase which senses variations in the extracellular milieu and a response regulator which alters gene expression upon activation by its cognate histidine kinase17 (Figure 1). TCS not only modulate virulence-associated mechanisms but also fundamental biological processes such as pathways involved in metabolism18,19 and osmoregulation20. The proportion of genes coding for TCS within a bacterial genome is thought to be dependent on a range of factors, including genome size and the diversity of different environments the bacterium may encounter21. Generally, histidine kinase and response regulator genes are co-transcribed in an operon, but they can also exist in the genome separated from their cognate partner, and are defined as orphans22. In A. baumannii, genomic analyses have identified 12 response regulator genes in an avirulent isolate that increases to 16 to 19 in pathogenic isolates23. Of these, only a handful have been experimentally examined24–29.
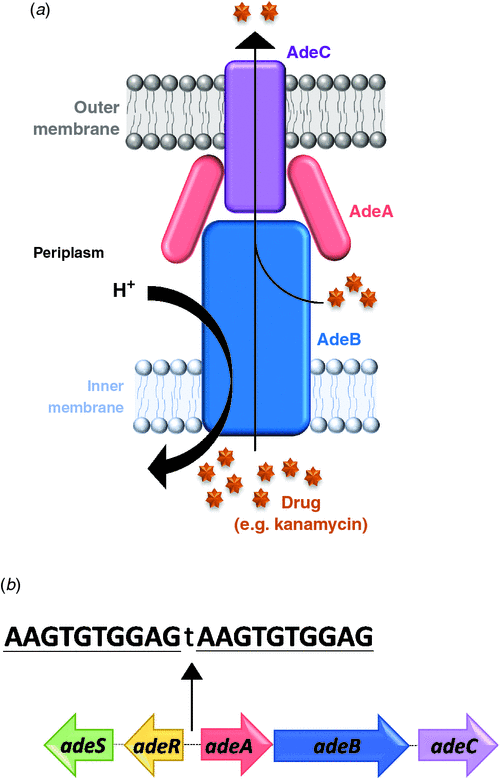
An aspect of my research has focused on the AdeRS TCS which was originally identified as a regulator of the AdeABC three component efflux system in many A. baumannii clinical isolates (Figure 2a). When overexpressed, AdeABC exports a broad range of structurally-unrelated antimicrobials including antibiotics, biocides and dyes25,30–34. Importantly, within this group of substrates are compounds from the carbapenem class of antibiotics as well as tigecycline, one of the last lines of defence against A. baumannii35. Analysis of many multidrug resistant A. baumannii isolates has shown a high incidence of mutations or the presence of insertional elements in the AdeRS regulatory system, deeming this TCS to be a significant contributor to the observed multidrug resistance phenotype11,36,37. At a genetic level, the adeRS genes lie adjacent to adeABC, but are divergently transcribed (Figure 2b). The AdeR response regulator protein binds to a 10 base-pair direct repeat DNA sequence and modulates adeABC expression38; however, the external signal(s) that interact with the AdeS histidine kinase remain unknown. Aside from changes in antimicrobial resistance, deletion of adeRS seen in some clinical isolates, has identified significant alterations in persistence strategies, such as the production of an extracellular protective matrix known as a biofilm32,33.
|
Many clinical A. baumannii isolates harbour different genetic arrangements of the adeRS and adeABC operons39. Examination into one A. baumannii clinical isolate identified that insertional-inactivation of the outer membrane component of the pump (AdeC) did not affect resistance towards two previously identified AdeABC substrates25. It was suggested that in the absence of AdeC, AdeAB can utilise an alternative outer membrane protein to form a functional tripartite complex25. Given that AdeC may not be essential to confer antimicrobial resistance, and the diverse genetic arrangements of these operons across clinical A. baumannii isolates, a key aim of my studies was to (1) ascertain whether clinical isolates which naturally lack adeC also confer antimicrobial resistance and to (2) determine if the previously observed regulatory properties governed by AdeRS are also maintained. The well characterised A. baumannii ‘type’ strain ATCC 1797840 isolated from an infant with fatal meningitis, which does not carry adeC, was chosen for genetic manipulation. Through double homologous recombination techniques, mutant derivatives targeting either adeRS or adeAB genes were generated and compared to the ATCC 17978 parent. Antibiogram analysis of the adeAB mutant identified changes in resistance to a subset of structurally related antimicrobials, including a commonly utilised clinical disinfectant. The role of the AdeRS TCS in modulating expression of adeABC is of current debate. As deletion of adeRS exhibited similar resistance levels to the adeAB deletion strain, my research supports the hypothesis that the AdeRS TCS activates expression of the adeAB(C) operon25. These research findings shed new light on the resistance capabilities of the AdeABC pump, questioning the views that AdeABC does not contribute towards the intrinsic resistance of A. baumannii and that antibacterial efflux can only occur when AdeABC is constitutively over-expressed32.
To identify the effects of the deletion of adeRS on the global transcriptional landscape, RNA-sequencing methodologies were employed. Numerous changes in gene transcription levels were identified including expression of adeAB. Additionally, other genes known to be important for virulence, such as iron sequestering and pilus assembly operons were differentially expressed. AdeR has previously been found to bind to a 10 base-pair direct repeat only found within the intercistronic region between the adeRS and adeABC operons38. Genomic analyses within ATCC 17978 also support this finding. Therefore, in the adeRS deletion strain, aside from alterations in adeAB expression, the transcriptional changes in the aforementioned virulence associated genes are likely to be indirect. Interestingly, these direct/indirect transcriptional changes differed from an adeRS deletion mutant constructed in a different A. baumannii clinical isolate33, emphasising that changes in the global transcriptional landscape are dependent on the isolate under investigation.
With a lack of currently effective antimicrobial treatments and a less than promising pipeline for the generation of new antibiotics, research into novel antimicrobial treatments is of significant interest. Histidine kinases of TCS are seen as attractive targets, primarily due to their presence in many pathogenic bacterial species16 and the absence of homologues in higher eukaryotes, including humans41. A number of novel inhibitors towards some TCS regulatory cascades present across a number of clinically relevant pathogenic bacterial species have been identified42; however, no inhibitors have been recognised for TCS found within A. baumannii. Results from this research area have made promising leads but progress is slow and many challenges still remain42.
In recent decades, A. baumannii has fast become an extremely problematic hospital-acquired pathogen, propelled by its ability to flourish within hostile clinical environments and accrue resistance to the current armamentarium of therapeutic treatments. The AdeABC efflux system is a known contributor to the multidrug resistance phenotype displayed by this organism. My research into this system has identified that AdeAB in ATCC 17978 is functional despite the absence of AdeC and can provide intrinsic antimicrobial resistance, albeit to a limited substrate range. Antibacterial drug research efforts over recent decades have highlighted the eligibility of targeting TCS regulatory cascades for the development as an alternate therapy to treat bacterial infections. In light of this research, the AdeRS system holds particular interest due to its direct role in regulating a key aspect of multidrug resistance in many clinical A. baumannii isolates. Further examination into the AdeRS TCS is required, particularly identifying the activating stimuli of this system. This knowledge may be instrumental in the identification of novel inhibitors, which could aid in the future treatment of infections caused by this formidable pathogen.
References
[1] Peleg, A.Y. et al. (2008) Acinetobacter baumannii: emergence of a successful pathogen. Clin. Microbiol. Rev. 21, 538–582.| Acinetobacter baumannii: emergence of a successful pathogen.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD1cXpslajs7Y%3D&md5=74da5326cb5664135b7e83d32249ae58CAS |
[2] Jones, R.N. (2010) Microbial etiologies of hospital-acquired bacterial pneumonia and ventilator-associated bacterial pneumonia. Clin. Infect. Dis. 51, S81–S87.
| Microbial etiologies of hospital-acquired bacterial pneumonia and ventilator-associated bacterial pneumonia.Crossref | GoogleScholarGoogle Scholar |
[3] Sievert, D.M. et al. (2013) Antimicrobial-resistant pathogens associated with healthcare-associated infections: summary of data reported to the National Healthcare Safety Network at the Centers for Disease Control and Prevention, 2009-2010. Infect. Control Hosp. Epidemiol. 34, 1–14.
| Antimicrobial-resistant pathogens associated with healthcare-associated infections: summary of data reported to the National Healthcare Safety Network at the Centers for Disease Control and Prevention, 2009-2010.Crossref | GoogleScholarGoogle Scholar |
[4] Wong, D. et al. (2017) Clinical and pathophysiological overview of Acinetobacter infections: a century of challenges. Clin. Microbiol. Rev. 30, 409–447.
[5] Anstey, N.M. et al. (2002) Community-acquired bacteremic Acinetobacter pneumonia in tropical Australia is caused by diverse strains of Acinetobacter baumannii, with carriage in the throat in at-risk groups. J. Clin. Microbiol. 40, 685–686.
| Community-acquired bacteremic Acinetobacter pneumonia in tropical Australia is caused by diverse strains of Acinetobacter baumannii, with carriage in the throat in at-risk groups.Crossref | GoogleScholarGoogle Scholar |
[6] Dexter, C. et al. (2015) Community-acquired Acinetobacter baumannii: clinical characteristics, epidemiology and pathogenesis. Expert Rev. Anti Infect. Ther. 13, 567–573.
| Community-acquired Acinetobacter baumannii: clinical characteristics, epidemiology and pathogenesis.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2MXlvV2qurs%3D&md5=90ba58b4ee40c8d96b10421e87927a77CAS |
[7] Vila, J. et al. (2007) Porins, efflux pumps and multidrug resistance in Acinetobacter baumannii. J. Antimicrob. Chemother. 59, 1210–1215.
| Porins, efflux pumps and multidrug resistance in Acinetobacter baumannii.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD2sXnvVahtLs%3D&md5=c7afee6ea2ef79c4cb0ddaacec8c2ba7CAS |
[8] Sugawara, E. and Nikaido, H. (2012) OmpA is the principal nonspecific slow porin of Acinetobacter baumannii. J. Bacteriol. 194, 4089–4096.
| OmpA is the principal nonspecific slow porin of Acinetobacter baumannii.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC38XhtV2htL7P&md5=58bca6b8c86a056bedf3ab2aac090907CAS |
[9] Fournier, P.E. et al. (2006) Comparative genomics of multidrug resistance in Acinetobacter baumannii. PLoS Genet. 2, e7.
| Comparative genomics of multidrug resistance in Acinetobacter baumannii.Crossref | GoogleScholarGoogle Scholar |
[10] Blackwell, G.A. et al. (2016) IncM Plasmid R1215 is the source of chromosomally located regions containing multiple antibiotic resistance genes in the globally disseminated Acinetobacter baumannii GC1 and GC2 clones. MSphere 1, e00117-16.
| IncM Plasmid R1215 is the source of chromosomally located regions containing multiple antibiotic resistance genes in the globally disseminated Acinetobacter baumannii GC1 and GC2 clones.Crossref | GoogleScholarGoogle Scholar |
[11] Wright, M.S. et al. (2017) Transcriptome remodeling of Acinetobacter baumannii during infection and treatment. MBio 8, e02193-16.
| Transcriptome remodeling of Acinetobacter baumannii during infection and treatment.Crossref | GoogleScholarGoogle Scholar |
[12] Yoon, E.J. et al. (2013) RND-type efflux pumps in multidrug-resistant clinical isolates of Acinetobacter baumannii: major role for AdeABC overexpression and AdeRS mutations. Antimicrob. Agents Chemother. 57, 2989–2995.
| RND-type efflux pumps in multidrug-resistant clinical isolates of Acinetobacter baumannii: major role for AdeABC overexpression and AdeRS mutations.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3sXhtVartrjK&md5=889ade8fa5092096bceeee31f54c9cc6CAS |
[13] Lemos, E.V. et al. (2014) Carbapenem resistance and mortality in patients with Acinetobacter baumannii infection: systematic review and meta-analysis. Clin. Microbiol. Infect. 20, 416–423.
| Carbapenem resistance and mortality in patients with Acinetobacter baumannii infection: systematic review and meta-analysis.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2cXptVaiu7c%3D&md5=1b26fdf6fe6d37c3cba9d69b551deb3aCAS |
[14] World Health Organization (2017) Global priority list of antibiotic-resistant bacteria to guide research, discovery, and development of new antibiotics. http://www.who.int/medicines/publications/global-priority-list-antibiotic-resistant-bacteria/en/
[15] Calva, E. and Oropeza, R. (2006) Two-component signal transduction systems, environmental signals, and virulence. Microb. Ecol. 51, 166–176.
| Two-component signal transduction systems, environmental signals, and virulence.Crossref | GoogleScholarGoogle Scholar | 1:STN:280:DC%2BD287hsVGmsA%3D%3D&md5=f075fcde1e29e17641d5dfb1d483e911CAS |
[16] Beier, D. and Gross, R. (2006) Regulation of bacterial virulence by two-component systems. Curr. Opin. Microbiol. 9, 143–152.
| Regulation of bacterial virulence by two-component systems.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD28XjtFagtLY%3D&md5=d6f6a603c26361577d7c9a885cad6115CAS |
[17] Casino, P. et al. (2010) The mechanism of signal transduction by two-component systems. Curr. Opin. Struct. Biol. 20, 763–771.
| The mechanism of signal transduction by two-component systems.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3cXhsV2js77K&md5=46c54541b56d25f9f08dd3cdb93ce219CAS |
[18] Kaspar, S. et al. (1999) The periplasmic domain of the histidine autokinase CitA functions as a highly specific citrate receptor. Mol. Microbiol. 33, 858–872.
| The periplasmic domain of the histidine autokinase CitA functions as a highly specific citrate receptor.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DyaK1MXls12hurk%3D&md5=d3c05d7583bad582b88f8113d6662c8aCAS |
[19] Zientz, E. et al. (1998) Fumarate regulation of gene expression in Escherichia coli by the DcuSR (dcuSR genes) two-component regulatory system. J. Bacteriol. 180, 5421–5425.
| 1:CAS:528:DyaK1cXmvVWitbc%3D&md5=719ee139d2421feeb2439051674761beCAS |
[20] Forst, S.A. and Roberts, D.L. (1994) Signal transduction by the EnvZ-OmpR phosphotransfer system in bacteria. Res. Microbiol. 145, 363–373.
| Signal transduction by the EnvZ-OmpR phosphotransfer system in bacteria.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DyaK2cXmvFKlt7o%3D&md5=66bdad8f9b8453a1a57f7fb709503f17CAS |
[21] Galperin, M.Y. (2005) A census of membrane-bound and intracellular signal transduction proteins in bacteria: bacterial IQ, extroverts and introverts. BMC Microbiol. 5, 35.
| A census of membrane-bound and intracellular signal transduction proteins in bacteria: bacterial IQ, extroverts and introverts.Crossref | GoogleScholarGoogle Scholar |
[22] Laub, M.T. et al. (2007) Phosphotransfer profiling: systematic mapping of two-component signal transduction pathways and phosphorelays. Methods Enzymol. 423, 531–548.
| Phosphotransfer profiling: systematic mapping of two-component signal transduction pathways and phosphorelays.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD1cXhtVOjtr8%3D&md5=6cae536341645cbd1a80a005f6672f42CAS |
[23] Adams, M.D. et al. (2008) Comparative genome sequence analysis of multidrug-resistant Acinetobacter baumannii. J. Bacteriol. 190, 8053–8064.
| Comparative genome sequence analysis of multidrug-resistant Acinetobacter baumannii.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD1cXhsV2lsLjM&md5=9e82c6d1a7e225e15ca98c9e6ec7f3b6CAS |
[24] Tomaras, A.P. et al. (2008) Characterisation of a two-component regulatory system from Acinetobacter baumannii that controls biofilm formation and cellular morphology. Microbiology 154, 3398–3409.
| Characterisation of a two-component regulatory system from Acinetobacter baumannii that controls biofilm formation and cellular morphology.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD1cXhsVCltbbF&md5=fcef43f6a6c3fe1493d7450e60623ae6CAS |
[25] Marchand, I. et al. (2004) Expression of the RND-type efflux pump AdeABC in Acinetobacter baumannii is regulated by the AdeRS two-component system. Antimicrob. Agents Chemother. 48, 3298–3304.
| Expression of the RND-type efflux pump AdeABC in Acinetobacter baumannii is regulated by the AdeRS two-component system.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD2cXnsVeiu7o%3D&md5=c3ed923bd4f597fab76c3072f26834e8CAS |
[26] Adams, M.D. et al. (2009) Resistance to colistin in Acinetobacter baumannii associated with mutations in the PmrAB two-component system. Antimicrob. Agents Chemother. 53, 3628–3634.
| Resistance to colistin in Acinetobacter baumannii associated with mutations in the PmrAB two-component system.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD1MXhtFequ7fK&md5=29abd19467977be95572eac3a9d58e55CAS |
[27] Cerqueira, G.M. et al. (2014) A global virulence regulator in Acinetobacter baumannii and its control of the phenylacetic acid catabolic pathway. J. Infect. Dis. 210, 46–55.
| A global virulence regulator in Acinetobacter baumannii and its control of the phenylacetic acid catabolic pathway.Crossref | GoogleScholarGoogle Scholar |
[28] Gebhardt, M.J. and Shuman, H.A. (2017) GigA and GigB are master regulators of antibiotic resistance, stress responses and virulence in Acinetobacter baumannii. J. Bacteriol. 199, e00066-17.
| GigA and GigB are master regulators of antibiotic resistance, stress responses and virulence in Acinetobacter baumannii.Crossref | GoogleScholarGoogle Scholar |
[29] Tipton, K.A. and Rather, P.N. (2017) An ompR/envZ two-component system ortholog regulates phase variation, osmotic tolerance, motility, and virulence in Acinetobacter baumannii strain AB5075. J. Bacteriol. 199, e00705-16.
| An ompR/envZ two-component system ortholog regulates phase variation, osmotic tolerance, motility, and virulence in Acinetobacter baumannii strain AB5075.Crossref | GoogleScholarGoogle Scholar |
[30] Magnet, S. et al. (2001) Resistance-nodulation-cell division-type efflux pump involved in aminoglycoside resistance in Acinetobacter baumannii strain BM4454. Antimicrob. Agents Chemother. 45, 3375–3380.
| Resistance-nodulation-cell division-type efflux pump involved in aminoglycoside resistance in Acinetobacter baumannii strain BM4454.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD3MXovVOls7o%3D&md5=cb3a016e9a1d4a0600254d9c18e63f9cCAS |
[31] Ruzin, A. et al. (2007) AdeABC multidrug efflux pump is associated with decreased susceptibility to tigecycline in Acinetobacter calcoaceticus-Acinetobacter baumannii complex. J. Antimicrob. Chemother. 59, 1001–1004.
| AdeABC multidrug efflux pump is associated with decreased susceptibility to tigecycline in Acinetobacter calcoaceticus-Acinetobacter baumannii complex.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD2sXmt1els74%3D&md5=b998028eef9cf3546c6eafa4c97c0f92CAS |
[32] Yoon, E.J. et al. (2015) Contribution of resistance-nodulation-cell division efflux systems to antibiotic resistance and biofilm formation in Acinetobacter baumannii. MBio 6, e00309-15.
| Contribution of resistance-nodulation-cell division efflux systems to antibiotic resistance and biofilm formation in Acinetobacter baumannii.Crossref | GoogleScholarGoogle Scholar |
[33] Richmond, G.E. et al. (2016) The Acinetobacter baumannii two-component system AdeRS regulates genes required for multidrug efflux, biofilm formation, and virulence in a strain-specific manner. MBio 7, e00430-16.
[34] Rajamohan, G. et al. (2010) Novel role of Acinetobacter baumannii RND efflux transporters in mediating decreased susceptibility to biocides. J. Antimicrob. Chemother. 65, 228–232.
| Novel role of Acinetobacter baumannii RND efflux transporters in mediating decreased susceptibility to biocides.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3cXhtVSqsL4%3D&md5=81bb567c741397b791c3919c2575abf8CAS |
[35] Doi, Y. et al. (2015) Acinetobacter baumannii: evolution of antimicrobial resistance-treatment options. Semin. Respir. Crit. Care Med. 36, 85–98.
| Acinetobacter baumannii: evolution of antimicrobial resistance-treatment options.Crossref | GoogleScholarGoogle Scholar |
[36] Hornsey, M. et al. (2010) AdeABC-mediated efflux and tigecycline MICs for epidemic clones of Acinetobacter baumannii. J. Antimicrob. Chemother. 65, 1589–1593.
| AdeABC-mediated efflux and tigecycline MICs for epidemic clones of Acinetobacter baumannii.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3cXptVyqtrg%3D&md5=dfbfff2b69182c0adc713d4cd13c2f14CAS |
[37] Sun, J.R. et al. (2012) A truncated AdeS kinase protein generated by ISAba1 insertion correlates with tigecycline resistance in Acinetobacter baumannii. PLoS One 7, e49534.
| A truncated AdeS kinase protein generated by ISAba1 insertion correlates with tigecycline resistance in Acinetobacter baumannii.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC38XhslyisL3L&md5=d383b9d2a00b594a4ae2b8c9979c7023CAS |
[38] Chang, T.Y. et al. (2016) AdeR protein regulates adeABC expression by binding to a direct-repeat motif in the intercistronic spacer. Microbiol. Res. 183, 60–67.
| AdeR protein regulates adeABC expression by binding to a direct-repeat motif in the intercistronic spacer.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2MXitVSgtr3M&md5=4cd551c528b74d3fafb1a9c0a1274014CAS |
[39] Nemec, A. et al. (2007) Relationship between the AdeABC efflux system gene content, netilmicin susceptibility and multidrug resistance in a genotypically diverse collection of Acinetobacter baumannii strains. J. Antimicrob. Chemother. 60, 483–489.
| Relationship between the AdeABC efflux system gene content, netilmicin susceptibility and multidrug resistance in a genotypically diverse collection of Acinetobacter baumannii strains.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD2sXpt1Cmur4%3D&md5=2bf0130f5c5b51c4f2be0720ff218429CAS |
[40] Smith, M.G. et al. (2007) New insights into Acinetobacter baumannii pathogenesis revealed by high-density pyrosequencing and transposon mutagenesis. Genes Dev. 21, 601–614.
| New insights into Acinetobacter baumannii pathogenesis revealed by high-density pyrosequencing and transposon mutagenesis.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD2sXjsVSqtbg%3D&md5=ada460e4d2d0a8d1c0aeb6cef095cc09CAS |
[41] Thomason, P. and Kay, R. (2000) Eukaryotic signal transduction via histidine-aspartate phosphorelay. J. Cell Sci. 113, 3141–3150.
| 1:CAS:528:DC%2BD3cXnsVyltL8%3D&md5=7d3015e01a3a6d0658fac14a36a4c90eCAS |
[42] Bem, A.E. et al. (2015) Bacterial histidine kinases as novel antibacterial drug targets. ACS Chem. Biol. 10, 213–224.
| Bacterial histidine kinases as novel antibacterial drug targets.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC2cXhvF2nt7jI&md5=c42fc79b89adfdd540aab0380a6f7705CAS |
Biography
Felise Adams is a PhD candidate from Flinders University, Adelaide, Australia, who is working under the supervision of Professor Melissa H Brown. Her research interests include molecular genetics, particularly gene regulation and antibiotic resistance strategies of pathogenic bacterial species.