Maternal nutrition and developmental programming of offspring
Lawrence P. Reynolds A * , Wellison J. S. Diniz B , Matthew S. Crouse C , Joel S. Caton A , Carl R. Dahlen A , Pawel P. Borowicz A and Alison K. Ward AA Center for Nutrition and Pregnancy, and Department of Animal Sciences, North Dakota State University, Fargo, ND 58108-6050, USA.
B Department of Animal Sciences, Auburn University, Auburn, AL 36832, USA.
C Nutrition, Growth, and Physiology Research Unit, USDA/Agricultural Research Service, U.S. Meat Animal Research Center, Clay Center, NE 68933-0166, USA.
Reproduction, Fertility and Development 35(2) 19-26 https://doi.org/10.1071/RD22234
Published online: 1 November 2022
© 2023 The Author(s) (or their employer(s)). Published by CSIRO Publishing on behalf of the IETS. This is an open access article distributed under the Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License (CC BY-NC-ND)
Abstract
Developmental programming is the concept that ‘stressors’ during development (i.e. pregnancy, the perinatal period and infancy) can cause long-term changes in gene expression, leading to altered organ structure and function. Such long-term changes are associated with an increased risk of a host of chronic pathologies, or non-communicable diseases including abnormal growth and body composition, behavioural or cognitive dysfunction, metabolic abnormalities, and cardiovascular, gastro-intestinal, immune, musculoskeletal and reproductive dysfunction. Maternal nutrition during the periconceptual period, pregnancy and postnatally can have profound influences on the developmental program. Animal models, including domestic livestock species, have been important for defining the mechanisms and consequences of developmental programming. One of the important observations is that maternal nutritional status and other maternal stressors (e.g. environmental temperature, high altitude, maternal age and breed, multiple fetuses, etc.) early in pregnancy and even periconceptually can affect not only embryonic/fetal development but also placental development. Indeed, altered placental function may underlie the effects of many maternal stressors on fetal growth and development. We suggest that future directions should focus on the consequences of developmental programming during the offspring’s life course and for subsequent generations. Other important future directions include evaluating interventions, such as strategic dietary supplementation, and also determining how we can take advantage of the positive, adaptive aspects of developmental programming.
Keywords: developmental programming, fetal programming, gene expression, maternal nutrition, organ function, organ structure, periconceptual, placental programming.
Introduction
Developmental programming, also known as fetal programming, is the concept that developmental stressors can alter gene expression in the developing fetus/neonate via epigenetic mechanisms. Such epigenetic alterations are thought to ‘program’ not only fetal and postnatal growth and development, but also to cause long-term changes in organ structure (e.g. altered number of nephrons in the kidney, altered pancreatic islet number or size, altered number of myofibres) and function (Barker 1990; Paneth and Susser 1995, 2004; Armitage et al. 2004; Wu et al. 2006; Caton and Hess 2010; Reynolds et al. 2010a, 2017, 2019, 2022; Reynolds and Caton 2012; Caton et al. 2019; Dahlen et al. 2021; Diniz et al. 2022). The consequences of developmental programming include preterm delivery, low birth weight and poor survival of newborns as well as a host of chronic pathologies in the offspring including abnormal growth and body composition, behavioural or cognitive dysfunction, metabolic abnormalities and cardiovascular, gastro-intestinal, immune, musculoskeletal and reproductive dysfunction (Barker 2004; Wu et al. 2006; Caton and Hess 2010; Reynolds et al. 2010a, 2017, 2019, 2022; Reynolds and Caton 2012; Reynolds and Vonnahme 2016; Caton et al. 2019; Cushman and Perry 2019; Dahlen et al. 2021). Additionally, chronically altered function of organ systems may contribute to aging and, importantly for livestock production, reduced longevity and production (Zambrano et al. 2014, 2015, 2020, 2021; Du et al. 2015; Franke et al. 2017; Kuo et al. 2017, 2018; Pankey et al. 2017; Yang et al. 2017; Broadhead et al. 2019; Cushman and Perry 2019; Wang et al. 2019).
The concept of developmental programming was first articulated by Dr. David Barker and colleagues. They used epidemiological evidence in humans to show a strong association between low birth weight, poor postnatal environment, or other developmental insults, and the subsequent risk of developing the range of pathologies noted above (Barker 1990, 2004). These studies suggested that an adverse intrauterine environment can lead to a greater incidence of non-communicable diseases in the offspring and that poor maternal nutrition was ‘.. an obvious suspect (Barker 1990, 2004).’ Subsequently, numerous epidemiological studies in humans, as well as many carefully controlled studies in various animal models, have confirmed the initial observations of Barker and colleagues, and some of the risk factors for developmental programming have been identified (Armitage et al. 2004; Wu et al. 2006; Caton and Hess 2010; Reynolds et al. 2010a, 2017, 2019, 2022; Reynolds and Caton 2012; Reynolds and Vonnahme 2017; Caton et al. 2019).
An additional, and important concept is that developmental programming must have entered and remained in the genome because of its adaptive advantages (Nettle et al. 2013; Bateson et al. 2014; Mueller et al. 2015). Whether we can take advantage of these adpatations to improve fitness and therefore productivity of the offspring is not known but should be kept in mind as we study various aspects of developmental programming (Reynolds et al. 2022).
Maternal nutrition
Maternal nutrition has a profound effect on fetal growth, as reflected by fetal weights in late pregnancy (Reynolds et al. 2019, 2022). For example, adolescent sheep that are overfed throughout pregnancy exhibit reduced fetal and placental weights similar to those of heat-stressed adult mothers. In this experimental paradigm, not only is placental vascular development reduced, but gravid uterine (maternal placental) and umbilical (fetal placental) blood flows are profoundly reduced, similar to that seen in heat-stressed pregnancies (Reynolds et al. 2019, 2022). Reduced placental blood flows are associated with altered placental vascular growth and development (Reynolds et al. 2006, 2010a, 2010b, 2019, 2022).
These observations are not surprising, as soon after implantation the placenta becomes the sole organ of exchange between the fetal and maternal systems. In other words, very early in pregnancy, the exchange of nutrients, oxygen and metabolic wastes between the maternal and fetal systems occurs solely via the placenta (Mayhew et al. 2004; Redmer et al. 2005, 2009; Vonnahme et al. 2007; Mayhew 2009; Reynolds et al. 2010b). Because it is primarily an organ of transport, the placenta also develops an extensive blood supply (i.e. it becomes highly vascular), which causes gravid uterine and umbilical blood flows (that represent the blood supplies to the maternal and fetal sides of the placenta, respectively) to increase exponentially throughout pregnancy. In fact, the increase in umbilical blood flow keeps pace with the exponential growth of the fetus (Reynolds and Redmer 1995; Reynolds et al. 2010b).
Many studies in livestock have shown that placental development and function are altered late in pregnancy in a variety of models of developmental programming, including maternal dietary intake (overfed or underfed), environmental heat stress, multiple pregnancies (singletons vs twins or triplets), maternal age (adolescent vs adult pregnancies), and maternal breed (Reynolds et al. 2019, 2022). These observations agree with those in other species, including humans (Mayhew et al. 2004; Reynolds et al. 2006; Mayhew 2009). In addition, defects in placental growth, including vascular defects, normally precede altered fetal growth and development (Reynolds et al. 2017, 2019).
We have shown in nutritionally-compromised pregnancies that placental vascular development is reduced very early in gestation (Grazul-Bilska et al. 2013, 2014; Vonnahme et al. 2013; Reynolds et al. 2014, 2019, 2022; Bairagi et al. 2016; McLean et al. 2017). Not only is placental vascular development altered by maternal nutrition during early pregnancy, but other aspects of placental function are affected as well. For example, placental expression of nutrient transporters and angiogenic factors is reduced in nutrient-restricted heifers during early pregnancy (Table 1). Similarly, maternal dietary restriction during early pregnancy profoundly affects gene expression in fetal liver, muscle and cerebrum (Table 1).
Because placental vascular growth and development are critical for normal placental function (Reynolds and Redmer 1995; Reynolds et al. 2010b, 2013, 2014; Bairagi et al. 2016), the observations described in the preceding paragraph have led to the concept of placental programming; i.e. that defects in placental growth and development underpin the altered fetal growth and organ development that is seen with developmental programming by maternal nutrition, and perhaps other ‘stressors’ such as in vitro production of embryos (see the next section of the manuscript; Reynolds et al. 2006; Vonnahme et al. 2013; Bairagi et al. 2016; Reynolds and Vonnahme 2017). Thus, despite the low metabolic demands of the conceptus in early pregnancy, these studies have confirmed that poor maternal diet can ‘program’ placental, and consequently fetal, development during their earliest stages (Diniz et al. 2021a, 2021b). In addition, maternal nutritional restriction is associated with effects, either directly or indirectly (e.g. via altered placental development and function) on gene expression in fetal organs (Table 1). Importantly, we also have shown that nutritional interventions (e.g. vitamin–mineral, energy and/or one-carbon metabolite supplementation; Table 1) can ‘rescue’ placental vascular development.
An additional time during which maternal nutrition seems to be important is the periconceptual period; that is, immediately before and after conception. Perhaps one of the most dramatic examples of developmental programming involves maternal nutrient restriction of ewes for 60 days before until 30 days after mating, which results in delivery an average of 7 days early compared with normally fed ewes and results in ‘… all preterm lambs [dying] soon after birth’ (Fig. 1; Bloomfield et al. 2003; Kumarasamy et al. 2005). Similarly, overfeeding or underfeeding of ewes for 8 weeks before collecting their oocytes results in poor rates of in vitro fertilisation and poor development of embryos in vitro (Fig. 2; Grazul-Bilska et al. 2012). These studies emphasise the importance of maternal nutritional status not only during early pregnancy but also during the periconceptual period.
|
|
|
|
As we have already mentioned, developmental programming must have entered and remained in the genome because of its adaptive advantages (Nettle et al. 2013; Bateson et al. 2014; Mueller et al. 2015), and taking advantage of these may improve fitness and therefore productivity of the offspring (Reynolds et al. 2022). For example, our studies of maternal nutritional status during the first 50 days of pregnancy in cattle showed that the vast majority of genes in fetal liver, muscle, and brain were upregulated in fetuses from nutrient-restricted dams (Table 1). Based on these observations, we have suggested that the upregulation of genes may represent an adaptive response to the maternal nutrient restriction (Crouse et al. 2019; Reynolds et al. 2022). This suggestion seems likely, as cattle under an extensive, pasture-based system, which is very common in regions with large areas of grassland such as the western region of the U.S., are often under nutritional stress during early pregnancy, which is typically in the fall, because of the declining yield and quality of pastures (Krysl et al. 1987; Wu et al. 2006; NASEM 2016; Caton et al. 2019, 2020). Thus, the adaptive response to limited maternal nutrient intake seems to involve upregulation of genes and even ‘rewiring’ of gene networks at the systems level (Caton et al. 2020; Diniz et al. 2021b).
In vitro production (IVP) of embryos
An instructive paradigm that is likely related to the nutritional, or perhaps more appropriately the environmental, status of the embryo during fertilisation and early development is that of in vitro production (IVP) of embryos. As shown by Loi et al. (2006) and subsequently many others, IVP by in vitro fertilisation or cloning results in a relatively low percentage (10–40%) of embryos surviving to term (Palmieri et al. 2008; Reynolds et al. 2013, 2014, 2019, 2022). Associated with the high rate of embryonic loss and poor pregnancy outcomes is poor embryonic and placental development very early in pregnancy (approximately 0.15, or 15%, of gestation; Arnold et al. 2006; Palmieri et al. 2007, 2008; Grazul-Bilska et al. 2013, 2014; Fidanza et al. 2014; Reynolds et al. 2014).
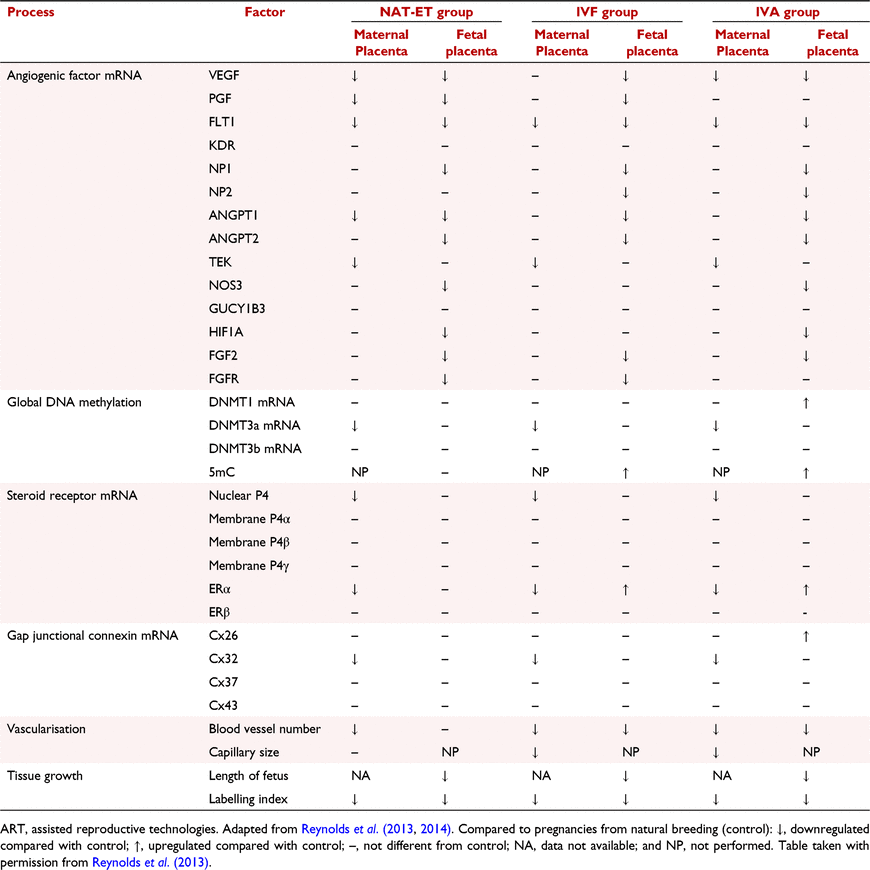
As shown in Table 2, poor embryonic and placental development after assisted reproductive technologies (ART) includes reduced fetal and placental growth; reduced placental vascularisation; reduced placental expression of a host of angiogenic factors, gap junctional connexins, and DNA methyl transferases; increased conceptus DNA methylation; and altered placental expression of oestrogen and progesterone receptors. All of these alterations would be expected to dramatically affect embryonic and placental growth and development and likely contribute to placental programming.
Observations in other mammalian species, including humans, have shown similar results after ART, including poor embryonic/fetal and placental development, poor pregnancy outcomes, and developmental programming of the offspring (Beaujean et al. 2004; Farin et al. 2006; Loi et al. 2006; Palmieri et al. 2007, 2008; Canovas et al. 2017; Coy et al. 2022). A promising approach to at least partially overcome the embryonic/fetal and placental defects seen with ART is the inclusion of oviductal or other reproductive fluids in the culture media. Inclusion of oviductal and other reproductive fluids improves the success of IVP and subsequent pregnancy rates and also reduces the effects of IVP on embryonic/fetal gene expression and DNA methylation status (Coy and Yanagimachi 2015; Canovas et al. 2017; Coy et al. 2022).
Conclusions and future directions
As discussed in this review, maternal nutritional status, ART, and a host of other maternal ‘stressors’ can profoundly affect embryonic/fetal development and survival, and often result in ‘programming’ of gene expression and subsequently altered growth and development. Recent data have shown that programming of placental growth and development very early in pregnancy may underlie many of the defects in embryonic/fetal growth and development that lead to poor outcomes during pregnancy and postnatally. In addition, maternal nutrition during the periconceptual period, that is, before and immediately after mating, also appears to program conceptus development.
Whether such early programming has long-lasting effects is unclear, but certainly epigenetic alterations occur, and there is every reason to believe they affect organ structure and function and will have lasting consequences throughout an individual’s life. Nevertheless, an important direction for future research is investigating the consequences of early programming both for health and productivity of the offspring and subsequent generations.
Another important area of future research focus should be whether interventions, including some of those discussed in this review, such as strategic dietary supplementation or culture of embryos with reproductive fluids, can mitigate some of the negative consequences of developmental programming, or even enhance the fitness of the offspring and future generations. That is, whether some of the strategies currently being evaluated for their ability to overcome the negative consequences of developmental programming can actually take advantage of the positive effects of developmental programming. This is especially apropos since, as we have already mentioned, it seems clear that we need a much better understanding of whether changes in expression of fetal and placental genes are adaptive, and therefore positive, or maladaptive, and therefore detrimental in both the short and long term.
Data availability
Any data from our research programs that are described in this review article will be shared upon reasonable request to the corresponding author.
Conflicts of interest
The authors declare no conflicts of interest.
Declaration of funding
Funding that supported the work from our research programs described in this manuscript came primarily from the Agriculture and Food Research Initiative (National Institute of Food and Agriculture, U.S. Department of Agriculture), the National Institutes of Health (U.S. Public Health Service), the North Dakota Agricultural Experiment Station, and the North Dakota State Board of Agricultural Research and Education.
Acknowledgements
The authors wish to acknowledge the many colleagues and collaborators, postdoctoral fellows, graduate and undergraduate students, and support staff who have made invaluable contributions to our work described in this manuscript.
References
Armitage, JA, Khan, IY, Taylor, PD, Nathanielsz, PW, and Poston, L (2004). Developmental programming of the metabolic syndrome by maternal nutritional imbalance: how strong is the evidence from experimental models in mammals? The Journal of Physiology 561, 355–377.| Developmental programming of the metabolic syndrome by maternal nutritional imbalance: how strong is the evidence from experimental models in mammals?Crossref | GoogleScholarGoogle Scholar |
Arnold, DR, Bordignon, V, Lefebvre, R, Murphy, BD, and Smith, LC (2006). Somatic cell nuclear transfer alters peri-implantation trophoblast differentiation in bovine embryos. Reproduction 132, 279–290.
| Somatic cell nuclear transfer alters peri-implantation trophoblast differentiation in bovine embryos.Crossref | GoogleScholarGoogle Scholar |
Bairagi, S, Quinn, KE, Crane, AR, Ashley, RL, Borowicz, PP, Caton, JS, Redden, RR, Grazul-Bilska, AT, and Reynolds, LP (2016). Maternal environment and placental vascularization in small ruminants. Theriogenology 86, 288–305.
| Maternal environment and placental vascularization in small ruminants.Crossref | GoogleScholarGoogle Scholar |
Barker, DJP (1990). The fetal and infant origins of adult disease. British Medical Journal 301, 1111.
| The fetal and infant origins of adult disease.Crossref | GoogleScholarGoogle Scholar |
Barker, DJP (2004). The developmental origins of well-being. Philosophical Transactions of the Royal Society of London. Series B: Biological Sciences 359, 1359–1366.
| The developmental origins of well-being.Crossref | GoogleScholarGoogle Scholar |
Bateson, P, Gluckman, P, and Hanson, M (2014). The biology of developmental plasticity and the predictive adaptive response hypothesis. The Journal of Physiology 592, 2357–2368.
| The biology of developmental plasticity and the predictive adaptive response hypothesis.Crossref | GoogleScholarGoogle Scholar |
Beaujean, N, Taylor, J, Gardner, J, Wilmut, I, Meehan, R, and Young, L (2004). Effect of limited DNA methylation reprogramming in the normal sheep embryo on somatic cell nuclear transfer. Biology of Reproduction 71, 185–193.
| Effect of limited DNA methylation reprogramming in the normal sheep embryo on somatic cell nuclear transfer.Crossref | GoogleScholarGoogle Scholar |
Bloomfield, FH, Oliver, MH, Hawkins, P, Campbell, M, Phillips, DJ, Gluckman, PD, Challis, JRG, and Harding, JE (2003). A periconceptional nutritional origin for noninfectious preterm birth. Science 300, 606.
| A periconceptional nutritional origin for noninfectious preterm birth.Crossref | GoogleScholarGoogle Scholar |
Broadhead, D, Mulliniks, JT, and Funston, RN (2019). Developmental programming in a beef production system. Veterinary Clinics of North America: Food Animal Practice 35, 379–390.
| Developmental programming in a beef production system.Crossref | GoogleScholarGoogle Scholar |
Canovas, S, Ivanova, E, Romar, R, García-Martínez, S, Soriano-Úbeda, C, García-Vázquez, FA, Saadeh, H, Andrews, S, Kelsey, G, and Coy, P (2017). DNA methylation and gene expression changes derived from assisted reproductive technologies can be decreased by reproductive fluids. eLife 6, e23670.
| DNA methylation and gene expression changes derived from assisted reproductive technologies can be decreased by reproductive fluids.Crossref | GoogleScholarGoogle Scholar |
Caton JS, Hess BW (2010) Maternal plane of nutrition: impacts on fetal outcomes and postnatal offspring responses. Invited Review. In ‘Proceedings of the 4th grazing livestock nutrition conference. (Eds BW Hess, T Delcurto, JGP Bowman, RC Waterman) pp. 104–122. (Western Section: American Society of Animal Science: Champaign, IL). Available at https://animal.ifas.ufl.edu/apps/dairymedia/rns/2011/12caton.pdf
Caton, JS, Crouse, MS, Reynolds, LP, Neville, TL, Dahlen, CR, Ward, AK, and Swanson, KC (2019). Maternal nutrition and programming of offspring energy requirements. Translational Animal Science 3, 976–990.
| Maternal nutrition and programming of offspring energy requirements.Crossref | GoogleScholarGoogle Scholar |
Caton, JS, Crouse, MS, McLean, KJ, Dahlen, CR, Ward, AK, Cushman, RA, Grazul-Bilska, AT, Neville, BW, Borowicz, PP, and Reynolds, LP (2020). Maternal periconceptual nutrition, early pregnancy, and developmental outcomes in beef cattle. Journal of Animal Science 98, skaa358.
| Maternal periconceptual nutrition, early pregnancy, and developmental outcomes in beef cattle.Crossref | GoogleScholarGoogle Scholar |
Coy, P, and Yanagimachi, R (2015). The common and species-specific roles of oviductal proteins in mammalian fertilization and embryo development. BioScience 65, 973–984.
| The common and species-specific roles of oviductal proteins in mammalian fertilization and embryo development.Crossref | GoogleScholarGoogle Scholar |
Coy, P, Romar, R, and Romero-Aguirregomezcorta, J (2022). The embryo culture media in the era of epigenetics: is it time to go back to nature? Animal Reproduction 19, e20210132.
| The embryo culture media in the era of epigenetics: is it time to go back to nature?Crossref | GoogleScholarGoogle Scholar |
Crouse, MS, Crouse, MS, McLean, KJ, Greseth, NP, Crosswhite, MR, Pereira, NN, Ward, AK, Reynolds, LP, Dahlen, CR, Neville, BW, Borowicz, PP, and Caton, JS (2017). Maternal nutrition and stage of early pregnancy in beef heifers: impacts on expression of glucose, fructose, and cationic amino acid transporters in utero-placental tissues. Journal of Animal Science 95, 5563–5572.
| Maternal nutrition and stage of early pregnancy in beef heifers: impacts on expression of glucose, fructose, and cationic amino acid transporters in utero-placental tissues.Crossref | GoogleScholarGoogle Scholar |
Crouse, MS, Caton, JS, Cushman, RA, McLean, KJ, Dahlen, CR, Borowicz, PP, Reynolds, LP, and Ward, AK (2019). Moderate nutrient restriction of beef heifers alters expression of genes associated with tissue metabolism, accretion, and function in fetal liver, muscle, and cerebrum by day 50 of gestation. Translational Animal Science 3, 855–866.
| Moderate nutrient restriction of beef heifers alters expression of genes associated with tissue metabolism, accretion, and function in fetal liver, muscle, and cerebrum by day 50 of gestation.Crossref | GoogleScholarGoogle Scholar |
Crouse, MS, McLean, KJ, Greseth, NP, Ward, AK, Reynolds, LP, Dahlen, CR, Neville, BW, Borowicz, PP, and Caton, JS (2020). The effects of maternal nutrient restriction and day of early pregnancy on the location and abundance of neutral amino acid transporters in beef heifer utero-placental tissues. Journal of Animal Science 98, skaa197.
| The effects of maternal nutrient restriction and day of early pregnancy on the location and abundance of neutral amino acid transporters in beef heifer utero-placental tissues.Crossref | GoogleScholarGoogle Scholar |
Crouse, MS, McLean, KJ, Dwamena, J, Neville, TL, Menezes, ACB, Ward, AK, Reynolds, LP, Dahlen, CR, Neville, BW, Borowicz, PP, and Caton, JS (2021). The effects of maternal nutrition during the first 50 d of gestation on the location and abundance of hexose and cationic amino acid transporters in beef heifer uteroplacental tissues. Journal of Animal Science 99, skaa386.
| The effects of maternal nutrition during the first 50 d of gestation on the location and abundance of hexose and cationic amino acid transporters in beef heifer uteroplacental tissues.Crossref | GoogleScholarGoogle Scholar |
Cushman, RA, and Perry, GA (2019). Developmental programming of fertility in livestock. Veterinary Clinics of North America: Food Animal Practice 35, 321–330.
| Developmental programming of fertility in livestock.Crossref | GoogleScholarGoogle Scholar |
Dahlen, CR, Borowicz, PP, Ward, AK, Caton, JS, Czernik, M, Palazzese, L, Loi, P, and Reynolds, LP (2021). Programming of embryonic development. International Journal of Molecular Sciences 22, 11668.
| Programming of embryonic development.Crossref | GoogleScholarGoogle Scholar |
Dávila Ruiz, BJ, Dahlen, CR, Hurlbert, JLL, Baumgaertner, F, Bochantin, KA, Menezes, ACB, Diniz, W, Kirsch, JD, Borowicz, PP, Cánovas, S, and Reynolds, LP (2022). PSV-B-17 Effect of dietary supplementation with vitamins/minerals and/or energy on fetoplacental vascularity in crossbred Angus heifers. Journal of Animal Science 100, 346–347.
| PSV-B-17 Effect of dietary supplementation with vitamins/minerals and/or energy on fetoplacental vascularity in crossbred Angus heifers.Crossref | GoogleScholarGoogle Scholar |
Diniz, WJS, Reynolds, LP, Borowicz, PP, Ward, AK, Sedivec, KK, McCarthy, KL, Kassetas, CJ, Baumgaertner, F, Kirsch, JD, Dorsam, ST, Neville, TL, Forcherio, JC, Scott, RR, Caton, JS, and Dahlen, CR (2021a). Maternal vitamin and mineral supplementation and rate of maternal weight gain affects placental expression of energy metabolism and transport-related genes. Genes 12, 385.
| Maternal vitamin and mineral supplementation and rate of maternal weight gain affects placental expression of energy metabolism and transport-related genes.Crossref | GoogleScholarGoogle Scholar |
Diniz, WJS, Crouse, MS, Cushman, RA, McLean, KJ, Caton, JS, Dahlen, CR, Reynolds, LP, and Ward, AK (2021b). Cerebrum, liver, and muscle regulatory networks uncover maternal nutrition effects in developmental programming of beef cattle during early pregnancy. Scientific Reports 11, 2771.
| Cerebrum, liver, and muscle regulatory networks uncover maternal nutrition effects in developmental programming of beef cattle during early pregnancy.Crossref | GoogleScholarGoogle Scholar |
Diniz, WJS, Crouse, MS, Caton, JS, Claycombe-Larson, KJ, Lindholm-Perry, AK, Reynolds, LP, Dahlen, CR, Borowicz, PP, and Ward, AK (2022). Data article: DNA methylation dataset of bovine embryonic fibroblast cells treated with epigenetic modifiers and divergent energy supply. Data in Brief 42, 108074.
| Data article: DNA methylation dataset of bovine embryonic fibroblast cells treated with epigenetic modifiers and divergent energy supply.Crossref | GoogleScholarGoogle Scholar |
Du, M, Wang, B, Fu, X, Yang, Q, and Zhu, M-J (2015). Fetal programming in meat production. Meat Science 109, 40–47.
| Fetal programming in meat production.Crossref | GoogleScholarGoogle Scholar |
Farin, PW, Piedrahita, JA, and Farin, CE (2006). Errors in development of fetuses and placentas from in vitro-produced bovine embryos. Theriogenology 65, 178–191.
| Errors in development of fetuses and placentas from in vitro-produced bovine embryos.Crossref | GoogleScholarGoogle Scholar |
Fidanza, A, Toschi, P, Zacchini, F, Czernik, M, Palmieri, C, Scapolo, P, Modlinski, JA, Loi, P, and Ptak, GE (2014). Impaired placental vasculogenesis compromises the growth of sheep embryos developed in vitro. Biology of Reproduction 91, 21.
| Impaired placental vasculogenesis compromises the growth of sheep embryos developed in vitro.Crossref | GoogleScholarGoogle Scholar |
Franke, K, Clarke, GD, Dahnke, R, Gaser, C, Kuo, AH, Li, C, Schwab, M, and Nathanielsz, PW (2017). Premature brain aging in baboons resulting from moderate fetal undernutrition. Frontiers in Aging Neuroscience 9, 92.
| Premature brain aging in baboons resulting from moderate fetal undernutrition.Crossref | GoogleScholarGoogle Scholar |
Grazul-Bilska, AT, Borowczyk, E, Bilski, JJ, Reynolds, LP, Redmer, DA, Caton, JS, and Vonnahme, KA (2012). Overfeeding and underfeeding have detrimental effects on oocyte quality measured by in vitro fertilization and early embryonic development in sheep. Domestic Animal Endocrinology 43, 289–298.
| Overfeeding and underfeeding have detrimental effects on oocyte quality measured by in vitro fertilization and early embryonic development in sheep.Crossref | GoogleScholarGoogle Scholar |
Grazul-Bilska, AT, Johnson, ML, Borowicz, PP, et al. (2014). Placental development during early pregnancy in sheep: effects of embryo origin on vascularization. Reproduction 147, 639–648.
| Placental development during early pregnancy in sheep: effects of embryo origin on vascularization.Crossref | GoogleScholarGoogle Scholar |
Grazul-Bilska, AT, Johnson, ML, Borowicz, PP, Baranko, L, Redmer, DA, and Reynolds, LP (2013). Placental development during early pregnancy in sheep: effects of embryo origin on fetal and placental growth and global methylation. Theriogenology 79, 94–102.
| Placental development during early pregnancy in sheep: effects of embryo origin on fetal and placental growth and global methylation.Crossref | GoogleScholarGoogle Scholar |
Krysl LJ, Galyean ML, Wallace JD, McCollum FT, Judkins MB, Branine ME, Caton JS (1987) ‘Cattle nutrition on blue grama rangeland in New Mexico.’ Bulletin 727. (New Mexico State University: Las Cruces, NM)
Kumarasamy, V, Mitchell, MD, Bloomfield, FH, Oliver, MH, Campbell, ME, Challis, JRG, and Harding, JE (2005). Effects of periconceptional undernutrition on the initiation of parturition in sheep. American Journal of Physiology - Regulatory, Integrative and Comparative Physiology 288, R67–R72.
| Effects of periconceptional undernutrition on the initiation of parturition in sheep.Crossref | GoogleScholarGoogle Scholar |
Kuo, AH, Li, C, Li, J, Huber, HF, Nathanielsz, PW, and Clarke, GD (2017). Cardiac remodelling in a baboon model of intrauterine growth restriction mimics accelerated ageing. The Journal of Physiology 595, 1093–1110.
| Cardiac remodelling in a baboon model of intrauterine growth restriction mimics accelerated ageing.Crossref | GoogleScholarGoogle Scholar |
Kuo, AH, Li, C, Huber, HF, Clarke, GD, and Nathanielsz, PW (2018). Intrauterine growth restriction results in persistent vascular mismatch in adulthood. The Journal of Physiology 596, 5777–5790.
| Intrauterine growth restriction results in persistent vascular mismatch in adulthood.Crossref | GoogleScholarGoogle Scholar |
Loi, P, Clinton, M, Vackova, I, Fulka, J, Feil, R, Palmieri, C, Salda, LD, and Ptak, G (2006). Placental abnormalities associated with post-natal mortality in sheep somatic cell clones. Theriogenology 65, 1110–1121.
| Placental abnormalities associated with post-natal mortality in sheep somatic cell clones.Crossref | GoogleScholarGoogle Scholar |
Mayhew, TM (2009). A stereological perspective on placental morphology in normal and complicated pregnancies. Journal of Anatomy 215, 77–90.
| A stereological perspective on placental morphology in normal and complicated pregnancies.Crossref | GoogleScholarGoogle Scholar |
Mayhew, TM, Wijesekara, J, Baker, PN, et al. (2004). Short communication: Morphometric evidence that villous development and fetoplacental angiogenesis are compromised by intrauterine growth restriction but not by pre-eclampsia. Placenta 25, 829–833.
| Short communication: Morphometric evidence that villous development and fetoplacental angiogenesis are compromised by intrauterine growth restriction but not by pre-eclampsia.Crossref | GoogleScholarGoogle Scholar |
McLean, KJ, Crouse, MS, Crosswhite, MR, Negrin Pereira, N, Dahlen, CR, Borowicz, PP, Reynolds, LP, Ward, AK, Neville, BW, and Caton, JS (2017). Impacts of maternal nutrition on uterine and placental vascularity and mRNA expression of angiogenic factors during the establishment of pregnancy in beef heifers. Translational Animal Science 1, 160–167.
| Impacts of maternal nutrition on uterine and placental vascularity and mRNA expression of angiogenic factors during the establishment of pregnancy in beef heifers.Crossref | GoogleScholarGoogle Scholar |
Mueller, CA, Eme, J, Burggren, WW, Roghair, RD, and Rundle, SD (2015). Challenges and opportunities in developmental integrative physiology. Comparative Biochemistry and Physiology Part A: Molecular & Integrative Physiology 184, 113–124.
| Challenges and opportunities in developmental integrative physiology.Crossref | GoogleScholarGoogle Scholar |
NASEM (2016) ‘Nutrient requirements of beef cattle.’ 8th revised edn. (The National Academies Press: Washington, DC) https://doi.org/10.17226/19014
Nettle, D, Frankenhuis, WE, and Rickard, IJ (2013). The evolution of predictive adaptive responses in human life history. Proceedings of the Royal Society B: Biological Sciences 280, 20131343.
| The evolution of predictive adaptive responses in human life history.Crossref | GoogleScholarGoogle Scholar |
Palmieri, C, Loi, P, Reynolds, LP, Ptak, G, and Della Salda, L (2007). Placental abnormalities in ovine somatic cell clones at term: a light and electron microscopic investigation. Placenta 28, 577–584.
| Placental abnormalities in ovine somatic cell clones at term: a light and electron microscopic investigation.Crossref | GoogleScholarGoogle Scholar |
Palmieri, C, Loi, P, Ptak, G, and Della Salda, L (2008). Review Paper: A review of the pathology of abnormal placentae of somatic cell nuclear transfer clone pregnancies in cattle, sheep, and mice. Veterinary Pathology 45, 865–880.
| Review Paper: A review of the pathology of abnormal placentae of somatic cell nuclear transfer clone pregnancies in cattle, sheep, and mice.Crossref | GoogleScholarGoogle Scholar |
Paneth, N, and Susser, M (1995). Early origin of coronary heart disease (the “Barker hypothesis”). BMJ 310, 411–412.
| Early origin of coronary heart disease (the “Barker hypothesis”).Crossref | GoogleScholarGoogle Scholar |
Pankey, CL, Walton, MW, Odhiambo, JF, Smith, AM, Ghnenis, AB, Nathanielsz, PW, and Ford, SP (2017). Intergenerational impact of maternal overnutrition and obesity throughout pregnancy in sheep on metabolic syndrome in grandsons and granddaughters. Domestic Animal Endocrinology 60, 67–74.
| Intergenerational impact of maternal overnutrition and obesity throughout pregnancy in sheep on metabolic syndrome in grandsons and granddaughters.Crossref | GoogleScholarGoogle Scholar |
Redmer, DA, Aitken, RP, Milne, JS, Reynolds, LP, and Wallace, JM (2005). Influence of maternal nutrition on messenger RNA expression of placental angiogenic factors and their receptors at midgestation in adolescent sheep. Biology of Reproduction 72, 1004–1009.
| Influence of maternal nutrition on messenger RNA expression of placental angiogenic factors and their receptors at midgestation in adolescent sheep.Crossref | GoogleScholarGoogle Scholar |
Redmer, DA, Luther, JS, Milne, JS, Aitken, RP, Johnson, ML, Borowicz, PP, Borowicz, MA, Reynolds, LP, and Wallace, JM (2009). Fetoplacental growth and vascular development in overnourished adolescent sheep at day 50, 90 and 130 of gestation. Reproduction 137, 749–757.
| Fetoplacental growth and vascular development in overnourished adolescent sheep at day 50, 90 and 130 of gestation.Crossref | GoogleScholarGoogle Scholar |
Reynolds, LP, and Caton, JS (2012). Role of the pre- and post-natal environment in developmental programming of health and productivity. Molecular and Cellular Endocrinology 354, 54–59.
| Role of the pre- and post-natal environment in developmental programming of health and productivity.Crossref | GoogleScholarGoogle Scholar |
Reynolds, LP, and Redmer, DA (1995). Utero-placental vascular development and placental function. Journal of Animal Science 73, 1839–1851.
| Utero-placental vascular development and placental function.Crossref | GoogleScholarGoogle Scholar |
Reynolds, LP, and Vonnahme, KA (2016). TRIENNIAL REPRODUCTION SYMPOSIUM: Developmental programming of fertility. Journal of Animal Science 94, 2699–2704.
| TRIENNIAL REPRODUCTION SYMPOSIUM: Developmental programming of fertility.Crossref | GoogleScholarGoogle Scholar |
Reynolds, LP, and Vonnahme, KA (2017). Livestock as models for developmental programming. Animal Frontiers 7, 12–17.
| Livestock as models for developmental programming.Crossref | GoogleScholarGoogle Scholar |
Reynolds, LP, Caton, JS, Redmer, DA, et al. (2006). Evidence for altered placental blood flow and vascularity in compromised pregnancies. The Journal of Physiology 572, 51–58.
| Evidence for altered placental blood flow and vascularity in compromised pregnancies.Crossref | GoogleScholarGoogle Scholar |
Reynolds, LP, Borowicz, PP, Caton, JS, Vonnahme, KA, Luther, JS, Hammer, CJ, Maddock Carlin, KR, Grazul-Bilska, AT, and Redmer, DA (2010a). Developmental programming: the concept, large animal models, and the key role of uteroplacental vascular development. Journal of Animal Science 88, E61–E72.
| Developmental programming: the concept, large animal models, and the key role of uteroplacental vascular development.Crossref | GoogleScholarGoogle Scholar |
Reynolds, LP, Borowicz, PP, Caton, JS, Vonnahme, KA, Luther, JS, Buchanan, DS, Hafez, SA, Grazul-Bilska, AT, and Redmer, DA (2010b). Uteroplacental vascular development and placental function: an update. The International Journal of Developmental Biology 54, 355–366.
| Uteroplacental vascular development and placental function: an update.Crossref | GoogleScholarGoogle Scholar |
Reynolds, LP, Vonnahme, KA, Lemley, CO, Redmer, DA, Grazul-Bilska, AT, Borowicz, PP, and Caton, JS (2013). Maternal stress and placental vascular function and remodeling. Current Vascular Pharmacology 11, 564–593.
| Maternal stress and placental vascular function and remodeling.Crossref | GoogleScholarGoogle Scholar |
Reynolds LP, Borowicz PP, Palmieri C, Grazul-Bilska AT (2014) Placental vascular defects in compromised pregnancies: effects of assisted reproductive technologies and other maternal stressors. In ‘Advances in fetal and neonatal physiology. Vol. 814’. Advances in Experimental Medicine and Biology. (Eds L Zhang, CA Ducsay) pp. 193–204. (Springer Science+Business Media: NY). https://doi.org/10.1007/978-1-4939-1031-1_17
Reynolds LP, Ward AK, Caton JS (2017) Epigenetics and developmental programming in ruminants – long-term impacts on growth and development. In ‘Biology of domestic animals’. (Eds CG Scanes, R Hill) pp. 85–121. (CRC Press/Taylor & Francis Group: Milton Park, UK)
Reynolds, LP, Borowicz, PP, Caton, JS, Crouse, MS, Dahlen, CR, and Ward, AK (2019). Developmental programming of fetal growth and development. Veterinary Clinics of North America: Food Animal Practice 35, 229–247.
| Developmental programming of fetal growth and development.Crossref | GoogleScholarGoogle Scholar |
Reynolds LP, McLean KJ, McCarthy KL, Diniz WJS, Menezes ACB, Forcherio JC, Scott RR, Borowicz PP, Ward AK, Dahlen CR, Caton JS (2022) Nutritional regulation of embryonic survival, growth and development. In ‘Recent advances in animal nutrition and metabolism. Vol. 1354’. Advances in Experimental Medicine and Biology. (Ed. G Wu) pp. 63–76. https://doi.org/10.1007/978-3-030-85686-1_4
Vonnahme, KA, Zhu, MJ, Borowicz, PP, Geary, TW, Hess, BW, Reynolds, LP, Caton, JS, Means, WJ, and Ford, SP (2007). Effect of early gestational undernutrition on angiogenic factor expression and vascularity in the bovine placentome. Journal of Animal Science 85, 2464–2472.
| Effect of early gestational undernutrition on angiogenic factor expression and vascularity in the bovine placentome.Crossref | GoogleScholarGoogle Scholar |
Vonnahme, KA, Lemley, CO, Shukla, P, and O’Rourke, ST (2013). 2011 and 2012 Early Careers Achievement Awards: Placental programming: how the maternal environment can impact placental function. Journal of Animal Science 91, 2467–2480.
| 2011 and 2012 Early Careers Achievement Awards: Placental programming: how the maternal environment can impact placental function.Crossref | GoogleScholarGoogle Scholar |
Wang, Q, Zhu, C, Sun, M, Maimaiti, R, Ford, SP, Nathanielsz, PW, Ren, J, and Guo, W (2019). Maternal obesity impairs fetal cardiomyocyte contractile function in sheep. The FASEB Journal 33, 2587–2598.
| Maternal obesity impairs fetal cardiomyocyte contractile function in sheep.Crossref | GoogleScholarGoogle Scholar |
Wu, G, Bazer, FW, Wallace, JM, and Spencer, TE (2006). Board-invited review: intrauterine growth retardation: implications for the animal sciences. Journal of Animal Science 84, 2316–2337.
| Board-invited review: intrauterine growth retardation: implications for the animal sciences.Crossref | GoogleScholarGoogle Scholar |
Yang, S, Gerow, KG, Huber, HF, Considine, MM, Li, C, Mattern, V, Comuzzie, AG, Ford, SP, and Nathanielsz, PW (2017). A decline in female baboon hypothalamo-pituitary-adrenal axis activity anticipates aging. Aging 9, 1375–1385.
| A decline in female baboon hypothalamo-pituitary-adrenal axis activity anticipates aging.Crossref | GoogleScholarGoogle Scholar |
Zambrano, E, Guzmán, C, Rodríguez-González, GL, Durand-Carbajal, M, and Nathanielsz, PW (2014). Fetal programming of sexual development and reproductive function. Molecular and Cellular Endocrinology 382, 538–549.
| Fetal programming of sexual development and reproductive function.Crossref | GoogleScholarGoogle Scholar |
Zambrano, E, Reyes-Castro, LA, and Nathanielsz, PW (2015). Aging, glucocorticoids and developmental programming. Age 37, 52.
| Aging, glucocorticoids and developmental programming.Crossref | GoogleScholarGoogle Scholar |
Zambrano, E, Reyes-Castro, LA, Rodríguez-González, GL, Chavira, R, and Nathanielsz, PW (2020). Aging endocrine and metabolic phenotypes are programmed by mother’s age at conception in a sex-dependent fashion in the rat. The Journals of Gerontology: Series A 75, 2304–2307.
| Aging endocrine and metabolic phenotypes are programmed by mother’s age at conception in a sex-dependent fashion in the rat.Crossref | GoogleScholarGoogle Scholar |
Zambrano, E, Nathanielsz, PW, and Rodríguez-González, GL (2021). Developmental programming and ageing of male reproductive function. European Journal of Clinical Investigation 51, e13637.
| Developmental programming and ageing of male reproductive function.Crossref | GoogleScholarGoogle Scholar |