279. A dynamic model of the control of pulsatile luteinizing hormone secretion by gonadotrophin-releasing hormone
T. R. Ferasyi A , H. Barrett B , D. Blache A and G. B. Martin AA School of Animal Biology, University of Western Australia, Crawley, WA, Australia
B Faculty of Medicine and Dentistry, University of Western Australia, Crawley, WA, Australia
Reproduction, Fertility and Development 17(9) 116-116 https://doi.org/10.1071/SRB05Abs279
Submitted: 26 July 2005 Accepted: 26 July 2005 Published: 5 September 2005
Abstract
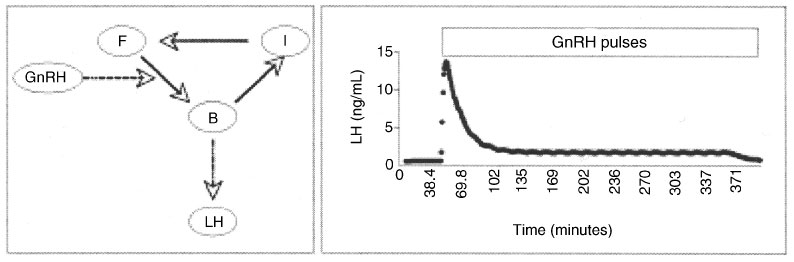
Infusion of GnRH in a continuous manner or with a very high pulse frequency initially stimulates but then downregulates LH secretion.1,2 This phenomenon is caused by the slow rate of internalisation of the GnRH receptor (GnRH-R) and the subsequent slow return of receptors to the plasma membrane of the gonadotroph.3 Pulsatile release of GnRH overcomes this problem by allowing a delay between successive stimulations. It is difficult to determine the relative importance of critical control points in this process in an animal model because GnRH activity reflects integrated inputs from many internal and external factors. We are therefore using SAAM II software to develop a compartmental model of the relationship between GnRH-R availability and LH responses following changes in GnRH pulse frequency. The model has three receptor states (free, bound, and internalised) and one LH compartment (Fig. 1). We assumed LH release is a function of the amount of receptor that binds GnRH. Following GnRH binding, receptors are rapidly lost as they enter the internalised state and then slowly returned to the membrane surface. We further assumed that the slow rate of receptor return explains the decrease in LH response with very high frequencies of GnRH pulses. The values for parameters were based on data obtained from experiments with sheep. In our current version of the model, downregulation is observed when gonadotrophs are stimulated with GnRH pulses every 15 min (Fig. 1), but not with pulses every 30 or 60 min, at a slow recycling rate (0.004 min–1). In contrast, LH secretion increases when GnRH is pulsed every 15 min and recycling rate is increased to 0.04 min–1. This suggests that, in sheep, a recycling rate between 0.004 and 0.04 min–1 is a critical aspect of the intracellular control of the process. Future work will include steroid feedback loops.