239 USE OF CYCLIC ADENOSINE MONOPHOSPHATE MODULATORS IN IN VITRO PRODUCTION OF BOVINE EMBRYOS
T. Fanti A , N. M. Ortega A , R. Garaguso A , M. J. Franco A , C. Herrera A and A. A. Mutto AInstituto de Investigaciones Biotecnológicas (IIB-INTECH), San Martín, Buenos Aires, Argentina
Reproduction, Fertility and Development 27(1) 209-209 https://doi.org/10.1071/RDv27n1Ab239
Published: 4 December 2014
Abstract
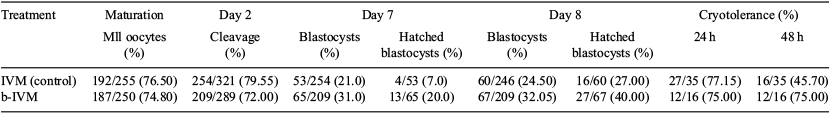
In vitro embryo production systems (IVP) try to emulate and enhance molecular events that occur in in vivo reproductive systems in order to increase, not only the number of embryos generated, but also their quality. Despite advances, IVP processes are still inefficient compared with in vivo systems. Several studies have attributed this deficiency to a lack of oocyte competence due to spontaneous premature resumption of meiotic maturation in the oocyte following the removal from its follicular environment. Therefore, our objective was to increase oocyte competence avoiding premature resumption of meiosis by using cyclic adenosine monophosphate modulators. Cumulus-oocyte complexes (COC) were obtained from ovaries of slaughterhouses, washed, and randomly allocated in 2 culture systems. Oocytes in the control group (IVM) were cultured for a period of 24 h in basal medium TCM-199 with EGF (1 µg mL–1) supplemented with rhFSH (25 mIU mL–1). Oocytes in the biphasic in vitro maturation (b-IVM) group were cultured for 2 h in a basal medium supplemented with a phosphodiesterase inhibitor, 3-isobutyl-1-methylxanthine (IBMX, 500 µM), and an activator of adenylate cyclase (forskolin, 100 µM). Subsequently, COC were washed and cultured in basal medium supplemented with cilostamide (20 µM) and rhFSH (25 mIU mL–1) for 24 h. Maturation rates were analysed and IVF was performed with a dose of 1 × 106 sperm cells mL–1 in IVF-SOF medium. The presumptive zygotes were cultured in continuous-single-culture medium (Irvine) supplemented with 8 mg mL–1 of BSA until they reached the blastocyst stage. No significant differences in maturation, cleavage, and cryotolerance were observed between b-IVM and IVM groups (P > 0.05; Table 1). This study showed that b-IVM produced a significant increase in IVP compared with the control (IVM) at Days 7 and 8 (P < 0.01). Blastocyst hatching rate was significant (P < 0.05) for both treatment and day of analysis. The b-IVM group yielded an increase of 10 and 7.5% at Days 7 and 8, respectively, of IVP. The biphasic maturation showed an improvement in quality regarding the control group, in the timing analysis of production, and hatching percentages, and these results show that the use of cyclic adenosine monophosphate modulators in the oocyte maturation process enhances oocyte competence, which is reflected in increased productivity and embryo quality. We propose this treatment as an alternative to the standard protocols currently used in IVP of bovine embryos.
|


