Does more maternal investment mean a larger brain? Evolutionary relationships between reproductive mode and brain size in chondrichthyans
Christopher G. Mull A C , Kara E. Yopak B and Nicholas K. Dulvy AA Earth to Ocean research group, Department of Biological Sciences, Simon Fraser University, Burnaby, BC, Canada.
B Center for Scientific Imaging, University of California, San Diego, La Jolla, CA, USA.
C Corresponding author. Email: cmull@sfu.ca
Marine and Freshwater Research 62(6) 567-575 https://doi.org/10.1071/MF10145
Submitted: 17 June 2010 Accepted: 25 February 2011 Published: 24 June 2011
Journal Compilation © CSIRO Publishing 2011 Open Access CC BY-NC-ND
Abstract
Chondrichthyans have the most diverse array of reproductive strategies of any vertebrate group, ranging from egg-laying to live-bearing with placental matrotrophy. Matrotrophy is defined as additional maternal provisioning beyond the yolk to the developing neonate; in chondrichthyans, this occurs through a range of mechanisms including uterine milk, oophagy, uterine cannibalism and placentotrophy. Chondrichthyans also exhibit a wide range of relative brain sizes and highly diverse patterns of brain organisation. Brains are energetically expensive to produce and maintain, and represent a major energetic constraint during early life in vertebrates. In mammals, more direct maternal–fetal placental connections have been associated with larger brains (steeper brain–body allometric scaling relationships). We test for a relationship between reproductive mode and relative brain size across 85 species from six major orders of chondrichthyans by using several phylogenetic comparative analyses. Ordinary least-squares (OLS) and reduced major axis (RMA) regression of body mass versus brain mass suggest that increased maternal investment results in a larger relative brain size. Our findings were supported by phylogenetic generalised least-squares models (pGLS), which also highlighted that these results vary with evolutionary tempo, as described by different branch-length assumptions. Across all analyses, maximum body size had a significant influence on the relative brain size, with large-bodied species (body mass >100 kg) having relatively smaller brains. The present study suggests that there may be a link between reproductive investment and relative brain size in chondrichthyans; however, a more definitive test requires a better-resolved phylogeny and a more nuanced categorisation of the level of maternal investment in chondrichthyans.
Additional keywords: allometry, encephalisation, pGLS, relative brain size, reproductive mode.
Introduction
The evolution of additional parental investment in offspring, such as through live-bearing and parental care, is thought to occur when the increased survival of offspring outweighs the parental costs of reduced fecundity and mobility (Clutton-Brock and Godfrey 1991; Reynolds et al. 2002). Though there have been numerous origins of parental care, there have been very few transitions to higher levels of maternal investment, such as placentation, in vertebrates (Wourms 1977; Wourms 1981; Dulvy and Reynolds 1997). Live-bearing is thought to have evolved more than 125 times in vertebrates (Shine 1989; Clutton-Brock and Godfrey 1991), with most transitions occurring in squamate reptiles and ∼9 or 10 transitions occurring in chondrichthyans (Dulvy and Reynolds 1997; Reynolds et al. 2002). The greatest number of gains and losses of maternal investment (matrotrophy) in vertebrates has occurred in the chondrichthyans, where matrotrophy has evolved four or five times and has potentially been lost 7–9 times (Dulvy and Reynolds 1997). In contrast, matrotrophy has evolved only four times in teleosts and appears never to have been lost (Wourms 1994; Goodwin et al. 2002). In fishes, including chondrichthyans, there appear to be few ecological correlates with reproductive mode; for example, there are no differences in the biogeographic distributions of live-bearers and egg-layers that cannot be explained by the differences in body size (Goodwin et al. 2005). However, there are life-history correlates with reproductive mode, with live-bearers having a large maternal body size, larger offspring size and lower fecundity (Goodwin et al. 2002). It still remains an open question as to why complex forms of reproductive investment have evolved in vertebrates (Shine 1989; Shine 2005; Reznick et al. 2007), particularly chondrichthyans.
The brain is perhaps the most energetically expensive organ to produce and maintain, and thus has been the subject of many allometric studies in mammals, birds and reptiles (Martin 1981, 1996; Iwaniuk and Nelson 2001; Elliot and Crespi 2008; Isler and van Schaik 2009). Most studies have focussed on functional implications of brain size and the development of major brain regions in vertebrates, examining ecological or behavioural correlates with brain organisation (Barton et al. 1995; Kotrschal et al. 1998; Lefebvre et al. 2002; Yopak et al. 2007; Shumway 2008), whereas few have examined any potential physiological mechanisms that drive this diversity (Elliot and Crespi 2008). Much of neural development occurs during gestation and brain tissue accounts for a disproportionate amount of metabolic costs in early life (Martin 1996; Elliot and Crespi 2008), raising questions about the role of reproductive diversity, and the level of maternal investment in the evolution of relative brain sizes.
Until recently, there has been little investigation into the diversity of reproductive strategies and modes of maternal investment in chondrichthyans and how they may correlate with the diverse brain morphologies documented across this group (Pagel and Harvey 1988; Martin 1996; Yopak et al. 2007). Previous studies of placental (eutherian) mammals suggest that it is worth searching for such a link; more ‘invasive’ forms of placentation (haemochorial), with the closest connection between maternal and fetal blood supply, exhibit a steeper brain–body allometric relationship than does less invasive placentation (endochorial and epithiliochorial) (Elliot and Crespi 2008). Hence, larger haemochorial mammals have larger brains for a given body size than do those with other less tightly connected placentas. Elliot and Crespi (2008) proposed that the development of larger brains is facilitated by better embryonic access to maternally derived fatty acids, which are essential for the development of brain tissue; however, whether this is true of cartilaginous fishes remains unknown.
Chondrichthyans, comprising more than 1100 extant sharks, skates, rays and chimaeras (Last 2007), offer an ideal taxon in which to study the potential links between reproduction and the brain size and morphology. Chondrichthyans have the most diverse array of reproductive strategies of any vertebrate taxa, ranging from single oviparity to placental viviparity, with many intermediate forms of matrotrophy (i.e. maternal nourishment provided to developing embryos) (Wourms 1977; Wourms and Demski 1993). The exact pattern of reproductive evolution in chondrichthyans remains open to debate, with older studies finding oviparity to be the ancestral form (Dulvy and Reynolds 1997), whereas studies using more recent phylogenetic hypotheses argue that viviparity is ancestral (Musick and Ellis 2005). Ultimately, the ancestral reproductive mode depends largely on the underlying evolutionary tree, which is still subject to considerable debate (Naylor et al. 2005; Vélez-Zuazo and Agnarsson 2010). Both Dulvy and Reynolds (1997) and Musick and Ellis (2005) yielded a relatively high amount of transitions between oviparity and viviparity; the former estimated 9 or 10 transitions from oviparity to viviparity, with two reversals, whereas the latter study estimated six transitions from viviparity to oviparity, with one reversal. The high number of transitions and reversals suggests that reproductive mode is evolutionarily labile. It is possible that this plasticity of reproductive mode has also developed as a result of changes in ecological roles, geographic distribution and the trade-offs associated with increased maternal investment (Goodwin et al. 2002, 2005; Crespi and Semeniuk 2004).
The relative brain size of chondrichthyans has been shown to be comparable to other vertebrates, with allometric-scaling ranges overlapping with reptiles, teleosts, birds and mammals (Bauchot et al. 1976; Northcutt 1977; Northcutt 1989). Much of the variation in brain organisation in chondrichthyans can be predicted by the overall brain size, with some major brain regions (e.g. telencephalon, cerebellum) enlarging disproportionately as the brain size increases (Yopak et al. 2010). Relative brain size and the relative development of major brain areas have been correlated with ecology (i.e. habitat type, feeding mode), and these patterns do not necessarily follow phylogenetic groupings (Yopak et al. 2007; Lisney et al. 2008; Yopak and Montgomery 2008; Yopak and Frank 2009), similar to patterns seen in other vertebrates, such as teleosts, birds and mammals (Huber et al. 1997; Kotrschal et al. 1998; de Winter and Oxnard 2001). There is some evidence that reproductive mode may be correlated with variation in brain size and organisation; the sharks with placental viviparity, such as the Carcharhinidae and Sphyrnidae, have among the largest relative brain sizes within chondrichthyans (Yopak et al. 2007), although this has not as yet been statistically tested.
Comparative analyses in mammals (Elliot and Crespi 2008; Barton and Capellini, 2011) suggest suggest that higher levels of maternal investment will increase nutrient allocation to developing embryos, allowing increased growth of energetically expensive brain tissue. Our primary hypothesis is that variation in the relative brain size among chondrichthyans is directly related to reproductive mode. Specifically, chondrichthyans with matrotrophy (additional maternal contribution beyond yolk sac, including the presence of histotroph, oophagy, or the formation of a yolk sac placenta), will exhibit larger relative brain sizes than do lecithotrophic (yolk-only nourishment) species.
Materials and methods
Data collection
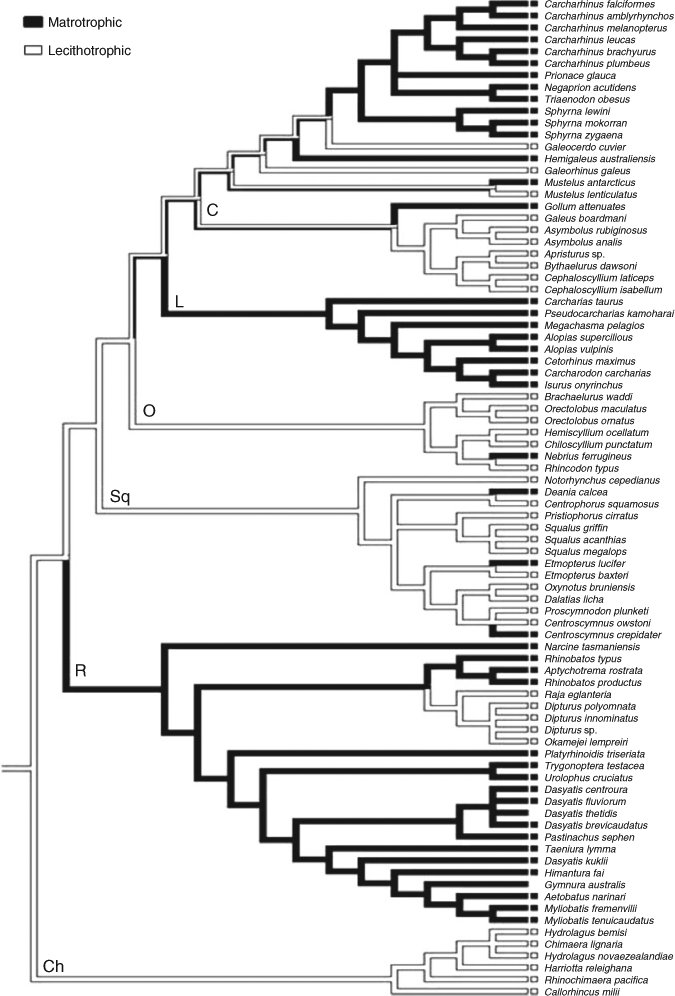
Data on brain weight and bodyweight for 85 chondrichthyan species were obtained from previously published studies (Northcutt 1977, 1978; Kruska 1988; Ito et al. 1999; Yopak et al. 2007; Lisney et al. 2008; Yopak and Montgomery 2008; Yopak and Frank 2009). Data on reproductive mode were collected from published literature (Dulvy and Reynolds 1997; Last and Stevens 2009) and each species was categorised into one of two reproductive modes, namely lecithotrophic or matrotrophic (Fig. 1).
Statistical analyses
To examine whether there was a difference in brain–body allometric scaling among reproductive modes, several general linear modelling methods were applied, including ordinary least-squares (OLS) and reduced major axis (RMA) regression. OLS regression is often used to examine changes in brain mass with body mass, and allometric-scaling relationships conform to the equation
where y = brain mass, x = body mass, a is the allometric coefficient and b is the allometric component. OLS regression assumes no error in the measurement of the independent variable (Ives et al. 2007), although this is often not the case with body-mass data. To account for inherent measurement error in the independent variable (i.e. body mass), we used reduced major axis (RMA) regression (Smith 2009). Despite the inherent statistical violations (particularly in the OLS method), we include them here to enable comparisons with previous research. For both regression methods, brain and body mass were log10-transformed and normality of the variables was confirmed with the Shapiro–Wilk test.
Because of shared evolutionary history, species are hierarchically autocorrelated and thus cannot be treated as independent samples drawn from a normal distribution (Freckleton 2000, 2009). Species are more likely to be similar to other species in the same genus because of their shared evolutionary history rather than because of convergent evolution to shared selective pressures. The use of OLS or RMA regression, without consideration of phylogenetic relationships, will often overestimate the extent of a correlation and result in Type I errors (Garland et al. 1992; Smith 2009). One way to account for this issue is to use a phylogeny to account for the relatedness of species by nesting the data. We tested the allometric relationship between brain mass and body mass by using phylogenetic generalised least-squares (pGLS) (Freckleton et al. 2002) as implemented in the APE package for R (Paradis 2006). This method accounts for the relatedness and hence correlation among species within families and families within order by accounting for the hypothesised phylogenetic relationships as a variance–covariance matrix in the generalised least-squares modelling framework.
A phylogenetic hypothesis of our study species was redrawn with the Mesquite phylogenetic analysis package (Maddison and Maddison 2010), using parsimony reconstruction. The tree used was primarily based on Shirai’s phylogeny (Shirai 1992, 1996) with additional information (Compagno 1988; Martin et al. 1992; Goto 2001; Rosenberger 2001; Didier 2004; McEachran and Aschliman 2004; Naylor et al. 2005) (Fig. 1). This parsimony tree provides only the topology (shape) of the relationships among species and as such does not provide any information on the evolutionary tempo or distance (as measured by branch lengths) among species. Hence, we considered two models of evolutionary distance among species. First, we considered two extreme transformations of a similar form, by setting branch lengths to either zero or one. Second, we considered a non-parametric rate-smoothing model, wherein rates change smoothly between connected branches of the tree (Sanderson 2002). This method was implemented using the ‘chronopl’ function, which produces longer branch-tip lengths, in the APE package in R (Paradis 2006). We tested the relative importance of reproductive mode for explaining the relative brain size by comparing two models using Akaike information criteria (AIC): ‘brain size ∼ body mass’ and ‘brain size ∼ body mass + reproductive mode’ (Hilborn and Mangel 1997; Burnham and Anderson 2002).
Following a preliminary analysis, data were reanalysed after the removal of large-bodied species (body mass >log10 5 or 100 kg: Prionace glauca, Sphyrna mokorran, Galeocerdo cuvier, Carcharias taurus, Isurus oxyrhinchus, Centorhinus maximus, Megachasma pelagios, Rhincodon typus, Carcharodon carcharias) to examine the potential effect of an extremely large body size on the relative brain size and the influence on the allometric coefficient (Striedter 2005).
Results
There is a positive relationship between brain mass and body mass, which appears to differ between the reproductive modes in a manner that suggests that increased maternal investment is associated with a larger relative brain size, but only in the smaller species (Fig. 2a). Matrotrophy results in brains that are 20–70% larger than those with lecithotrophy for chondrichthyans between 3 kg (log10 3.5) and 100 kg (log10 5). This marked difference in relative brains mass at small body mass (significant difference in intercept, P < 0.05) decays with increasing body size (there is no significant difference between slopes; P = 0.3382) (Table 1). This pattern is more striking if the largest chondrichthyans (>100 kg, log10 body mass of >5) are removed from the statistical and graphical analysis (Fig. 2b). The smallest matrotrophic species have brain masses that are on average 70% larger (difference between intercepts significant at P = 0.0225) and this advantage increases for sharks up to 100 kg, where relative brain sizes are 87% larger for matrotrophic species (a significant difference between slopes; P <0.0001) (Table 1). This marked difference with the exclusion of large-bodied species indicates that the mode of body-size evolution may have a significant effect on relative brain sizes, a phenomenon known as ‘gigantism’ (Striedter 2005). This pattern is robust to statistical method and the incorporation of phylogenetic information.

|
Reduced major axis regression of all body sizes yielded similar results, with no significant (P = 0.962) difference between the slopes, and a significant difference between the intercepts (P < 0.001) (Table 1). When all body sizes were included, matrotrophic species exhibited brains that were 6–26? larger than those of lecithotrophic species (Fig. 2c). When large body sizes (body mass >100 kg) were removed, there was a significant difference in both slope (P = 0.007) and intercept (P < 0.001) (Table 1). Similar to OLS regression, differences in relative brain size were more pronounced when large body sizes were excluded, with matrotrophy resulting in brains that were 55–68% larger than those of lecithotrophic species (Fig. 2d).
The phylogenetic GLS results were highly dependent on the branch-length assumptions of the available tree. The uniform branch-length transformation (zero or one) suggests that the inclusion of reproductive mode was not significant when all body sizes were included (Table 2). The use of a smoothed branch-length transformation yielded a more parsimonious model that included reproductive mode as a significant factor for all body sizes (Table 2). When large-bodied species (body mass >100 kg) were removed, the inclusion of reproductive mode improved all models irrespective of tree topology (Table 2). The finding of a robust signal despite tree topology suggests that although increased forms of maternal investment are associated with a larger relative brain size, body-size evolutionary trends may exert a significant influence on the brain development.
Discussion
The present study represents the first test of whether maternal investment beyond the yolk may contribute significantly to the relative brain size of chondrichthyans. Our results provide intriguing evidence that increased levels of maternal input during gestation through matrotrophy may facilitate the development of relatively larger brains in these species, particularly for smaller species (body mass <100 kg). This pattern is consistent across statistical methods; however, it is most pronounced at smaller body sizes, suggesting that the reproductive mode and maximum body size exert a significant influence on the relative brain size.
Encephalisation and reproduction
Across vertebrate taxa, small-bodied species tend to have larger relative brain sizes than do larger-bodied species, potentially driven by metabolic constraints and body-size evolution (Striedter 2005). Relative brain sizes of small-bodied (body mass <100 kg) matrotrophic species are larger than those of small-bodied lecithotrophic species, although this difference breaks down for larger body sizes (body mass >100 kg). This observed pattern is potentially explained by ‘gigantism’, where dramatic evolutionary increases in body size often do not have concurrent increases in the absolute brain size, resulting in smaller relative brains for larger-bodied species (Striedter 2005). This is especially pronounced in matrotrophic species (7 of 9 large-bodied species in the present study), where relative brain size appears to decline rapidly at body mass >100 kg. It is important to note, however, that the neonatal body size is not always correlated with maximum adult body size. The lecithotrophic whale shark (R. typus) is the largest extant fish, reaching a maximum size of 20 m total length (TL), and has pups of ∼0.5 m TL, whereas the matrotrophic white shark (C. carcharias) has pups >1 m TL. These were the two largest species included in the present dataset, and individuals had very similar brain and body masses, yet data may have been collected from very different life-history stages. Interspecific differences in body size can be reversed between neonate and adult life stages in chondrichthyans and this must be accounted for in data collection. One confounding factor of the present analysis is the varying life stages of samples used. To tease out the effect of maternal investment on brain development, future studies will need to examine differences, specifically in neonatal brain–body allometry.
The large relative brain sizes of small-bodied matrotrophic species is of note from a reproductive stand point, as many of these species are from the Order Carcharhiniformes and exhibit placental viviparity. Species from Carcharhiniformes, especially those from Sphyrnidae and Carcharhinidae, exhibit larger brains than expected, on the basis of allometric scaling of raw species data, significantly larger than do lecithotrophic species of a similar body size. With a few exceptions, members of Carcharhinidae and Sphyrnidae have among the most highly encephalised brains (Yopak et al. 2007), suggesting an influence of a more direct maternal fetal connection that has been associated with increased relative brain size in mammals (Martin 1996; Elliot and Crespi 2008). Developing embryos of placental species have more direct access to maternal resources, specifically to long-chain fatty acids, which are important for the development of neural tissue (Elliot and Crespi 2008). Thus, whereas lecithotrophic species are limited in their supply of resources during development (i.e. yolk sac), matrotrophic species have access to resources limited only by the mother’s energetic resources.
Variation in brain size is likely to be better explained with a more nuanced measuring of maternal investment and inclusion of other reproductive parameters (i.e. litter size, gestation or incubation length). In chondrichthyans, the relative size of most major brain areas, including the telencephalon and cerebellum, are highly predictable from the overall brain size (Yopak et al. 2007, 2010; Lisney et al. 2008; Yopak and Montgomery 2008), potentially owing to a conserved order of neurogenesis (Yopak et al. 2010), as documented in other vertebrates (Finlay and Darlington 1995; Finlay et al. 1998, 2001). The telencephalon and cerebellum, in particular, enlarge disproportionately as the absolute brain size increases in chondrichthyans and scale similarly to the neocortex and cerebellum of mammals (Yopak et al. 2010), brain areas that have been shown to continue neurogenesis longest through early development in mammals (Finlay and Darlington 1995; Yopak et al. 2010). This suggests that maternal investment may not be as simple as the amount of energy allocated during development, but instead may include the amount of time over which resources are allocated, and the window of time over which neurogenesis occurs. With gestation lengths ranging from 3 to 22 months (Wourms 1977; Wourms and Demski 1993), chondrichthyans are an ideal taxon for examining these hypotheses.
Life-history strategies and relative investment
There are other life-history parameters correlated with body size in chondrichthyans, which could exert some influence on the relative offspring brain size, such as litter size and gestation period, a pattern similarly observed in mammals (Pagel and Harvey 1988). In sharks, litter size is positively correlated with the maximum body size of the species, a relationship that varies with the reproductive mode (Cortés 2000; Goodwin et al. 2002). There is a trade-off between the size and number of offspring, and larger sharks tend to have smaller offspring than do smaller-bodied species, after litter size is accounted for (Smith and Fretwell 1974; Cortés 2000; Goodwin et al. 2002). With fewer embryos to nourish, smaller-bodied sharks can potentially allocate proportionally more resources to each individual embryo, enhancing brain growth per individual at the cost of overall fecundity for the mother. The benefits of per-offspring investment, such as larger neonate brain sizes, may well be greater for mothers of small-bodied species (Smith and Fretwell 1974) and, indeed, there may be a greater selection for precociality in the neonates of smaller species because of the elevated risk of predation on small individuals (Branstetter 1990).
The trade-off between the litter size and pup size can also be significant, and sharks show an inverse correlation between the litter size and offspring size (Cortés 2000). Lecithotrophic species exhibit a wide range of fecundities, from 2 to 300 pups per litter (Cortés 2000), and species with the largest litter sizes tended to have the lowest relative brain sizes (i.e. Rhincodon typus, Notorhynchus cepedianus) (Yopak et al. 2007; Yopak and Frank 2009). Indeed, in the construction of the pGLSs to explain apparent differences in the relative brain size, the best-fit models according to AIC criterion included both gestation length and maximum litter size (C. Mull, unpubl. data), although neither of these factors exerted a significant effect alone, which suggests a potentially fruitful avenue for further research. Although both lecithotrophic and matrotrophic species exhibited significant allometric-scaling coefficients, more variability in these linear models could potentially be explained with the inclusion of other life-history and reproductive parameters, although these are not yet available for all species.
We used a simple binary categorisation of reproductive mode into lecithotrophy and matrotrophy, which does not account for variation in the degree of maternal investment among species (Wourms and Lombardi 1992). One way of measuring the degree of relative investment in offspring is to consider the relative mass increase between ovum and neonate size. Many lecithotrophic species produce offspring that are considerably lighter than the ovum mass, likely as a result of the loss of energy because of metabolic conversion during development (Hou et al. 2008). For example, there is a 21% and 40% reduction in dry mass between the ovum and the neonate stage in the egg-laying (oviparous) lesser-spotted dogfish (Scyliorhinus canicula) and the lecithotrophic piked dogfish (Squalus acanthias) (Wourms 1993). By contrast, in matrotrophic species, there is a 1286% and 6 806 169% increase in neonate dry mass compared with the dry ovum mass in the pelagic stingray (Pteroplatytrygon violacea), which exhibits uterine milk and embryonic vilification, and the sandtiger shark (Odontaspis taurus) which exhibits oophagy and intra-uterine cannibalism (adelphophagy), respectively (Wourms 1993). One of the greatest levels of degree of placentotrophic maternal investment is exhibited by a small-bodied spadenose shark (Scoliodon laticaudus), which exhibits as 5 833 845% mass increases from ovum to neonate (Wourms 1993).
Future directions
The tentative conclusions we draw from the pGLS analysis highlight the need for a more highly resolved phylogeny of chondrichthyans that includes branch lengths. The use of trees with different branch lengths yielded different results, with some including the reproductive mode as a significant factor affecting brain mass. The development of broad chondrichthyan molecular phylogenies will contribute significantly to more powerful tests for potential linkages between maternal investment and brain size.
Aside from awaiting better phylogenetic hypotheses, we suggest the following three directions for future research in the area of maternal investment, brain size and brain morphology: (1) an increased sample size is needed to adequately represent and provide statistical rigour across every reproductive mode, (2) future models of allometric scaling should consider other reproductive parameters (i.e. fecundity, gestation length) as those data become available, and (3) finally, future sampling must also focus on neonatal and juvenile individuals to adequately measure any potential effects of maternal investment.
Conclusions
There are allometric-scaling differences between reproductive modes in chondrichthyans, with matrotrophic species exhibiting a positive grade shift in regard to their allometric-scaling relationship. Smaller-bodied species with matrotrophic reproductive modes tend to have larger relative brain sizes, whereas larger-bodied species show no differences between the modes. This suggests that additional maternal investment may have evolved to provide offspring with a ‘head-start’ in brain development, particularly for small-bodied species, but that, ultimately, the maximum body size may be a limiting factor on relative adult brain size. Further data collection and analysis will allow researchers to examine the energetic relationship of the maternal–fetal conflict in greater detail, and estimate the amount of caloric input per pup and how this correlates with the relative brain size, as well as accounting for other influencing factors such as litter size and gestation. The present study represents a first examination of the evolutionary linkages between the level of maternal investment and relative brain size in chondrichthyan fishes, and will hopefully provide a platform on which new studies can be built in the future.
Acknowledgements
We thank B. Crespi and A. Mooers for their valuable suggestions with the study, and W. Stein for his valuable contributions with pGLSs. We thank the guest editors and anonymous referees for their insightful comments and suggestions that helped greatly improve this manuscript. We thank the Sharks International Organising Committee for the student travel grant and inclusion of this manuscript in the proceedings. We also thank NSERC and the SFU graduate fellowship for funding in support of this research.
References
Barton, R. A., and Capellini, I. (2011). Maternal investment, life histories, and the costs of brain growth in mammals. Proceedings of the National Academy of Sciences 108, 6169–6174.| 1:CAS:528:DC%2BC3MXltVymu74%3D&md5=01e49d538d53c936b65df6f02dc4a0b3CAS |
Barton, R. A., Purvis, A., and Harvey, P. H. (1995). Evolutionary radiation of visual and olfactory brain systems in primates, bats and insectivores. Philosophical Transactions of the Royal Society of London. Series B. Biological Sciences 348, 381–392.
| Evolutionary radiation of visual and olfactory brain systems in primates, bats and insectivores.Crossref | GoogleScholarGoogle Scholar | 1:STN:280:DyaK28%2Fot1Onsg%3D%3D&md5=a388c7715b52f5bb5de67d3621237b09CAS |
Bauchot, R., Platel, R., and Ridet, J. M. (1976). Brain–body weight relationships in selachii. Copeia , 305–310.
| Brain–body weight relationships in selachii.Crossref | GoogleScholarGoogle Scholar |
Branstetter, S. (1990). Early life-history implications of selected charcharhind and lamnoid sharks of the northwest Atlantic. In ‘Elasmobranchs and Living Resources: Advances in the Biology, Ecology, Systematics and the Status of the Fisheries’. (Eds H. L. Pratt, S. H. Gruber and T. Taniuchi.) pp. 17–82. US Department of Commerce, NOAA Technical Report NMFS 90.
Burnham, K., and Anderson, D. (2002). ‘Model Selection and Multimodel Inference: A Practical Information Theoretic Approach.’ (Springer-Verlag: New York.)
Clutton-Brock, T. H., and Godfrey, C. (1991). Parental Investment. In ‘Behavioral Ecology: An Evolutionary Approach’. (Eds J. R. Krebs and N. B. Dabies.) pp. 234–262. (Cambridge University Press: Cambridge, UK.)
Compagno, L. (1988). ‘Sharks of the Order Carcharhiniformes.’ (Princeton University Press: Princeton, NJ.)
Cortés, E. (2000). Life history patterns and correlations in sharks. Reviews in Fisheries Science 8, 299–344.
Crespi, B., and Semeniuk, C. (2004). Parent-offspring conflict in the evolution of vertebrate reproductive mode. American Naturalist 163, 635–653.
| Parent-offspring conflict in the evolution of vertebrate reproductive mode.Crossref | GoogleScholarGoogle Scholar | 15122484PubMed |
de Winter, W., and Oxnard, C. E. (2001). Evolutionary radiations and convergences in the structural organization of mammalian brains. Nature 409, 710–714.
| Evolutionary radiations and convergences in the structural organization of mammalian brains.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD3MXht1yntrY%3D&md5=da39902b5e751867afb9676237ba1bbdCAS | 11217859PubMed |
Didier, D. A. (1995). ‘Phylogenetic Systematics of Extant Chimaeroid Fishes (Holocephali, Chimaeroidei)’, No. 3119. New York, NY: American Museum of Natural History.
Didier, D. (2004). Phylogeny and classification of extant Holocephali. In ‘Biology of Sharks and Their Relatives’. (Eds J. Carrier, J. Musick and M. Heithaus.) pp. 115–135. (CRC Press: New York.)
Dulvy, N. K., and Reynolds, J. D. (1997). Evolutionary transitions among egg-laying, live-bearing and maternal inputs in sharks and rays. Proceedings. Biological Sciences 264, 1309–1315.
| Evolutionary transitions among egg-laying, live-bearing and maternal inputs in sharks and rays.Crossref | GoogleScholarGoogle Scholar |
Elliot, M. G., and Crespi, B. J. (2008). Placental invasiveness and brain–body allometry in eutherian mammals. Journal of Evolutionary Biology 21, 1763–1778.
| Placental invasiveness and brain–body allometry in eutherian mammals.Crossref | GoogleScholarGoogle Scholar | 1:STN:280:DC%2BD1cjkvFyhtw%3D%3D&md5=a5d8201567116d7ff45f980582864f71CAS | 18808441PubMed |
Finlay, B. L., and Darlington, R. B. (1995). Linked regularities in the development and evolution of mammalian brains. Science 268, 1578–1584.
| Linked regularities in the development and evolution of mammalian brains.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DyaK2MXmtlarsb8%3D&md5=14cd9ffe6b17285736a11cc923019225CAS | 7777856PubMed |
Finlay, B. L., Hersman, M. N., and Darlington, R. B. (1998). Patterns of vertebrate neurogenesis and the paths of vertebrate evolution. Brain, Behavior and Evolution 52, 232–242.
| Patterns of vertebrate neurogenesis and the paths of vertebrate evolution.Crossref | GoogleScholarGoogle Scholar | 1:STN:280:DyaK1M%2FgsFeqtA%3D%3D&md5=7a14f43090d63d201d4a3ee03d87179bCAS | 9787222PubMed |
Finlay, B. L., Darlington, R. B., and Nicastro, N. (2001). Developmental structure in brain evolution. Behavioral and Brain Sciences 24, 298–308.
Freckleton, R. P. (2000). Phylogenetic tests of ecological and evolutionary hypotheses: checking for phylogenetic independence. Functional Ecology 14, 129–134.
| Phylogenetic tests of ecological and evolutionary hypotheses: checking for phylogenetic independence.Crossref | GoogleScholarGoogle Scholar |
Freckleton, R. P. (2009). The seven deadly sins of comparative analysis. Journal of Evolutionary Biology 22, 1367–1375.
| The seven deadly sins of comparative analysis.Crossref | GoogleScholarGoogle Scholar | 1:STN:280:DC%2BD1MvkvVegsQ%3D%3D&md5=ff481dd2263fb6b6449678ab036fb643CAS | 19508410PubMed |
Freckleton, R. P., Harvey, P. H., and Pagel, M. (2002). Phylogenetic analysis and comparative data: a test and review of evidence. American Naturalist 160, 712–726.
| Phylogenetic analysis and comparative data: a test and review of evidence.Crossref | GoogleScholarGoogle Scholar | 1:STN:280:DC%2BD1critl2ltw%3D%3D&md5=7f1301d9dac725b1eb19473a4f381e62CAS | 18707460PubMed |
Garland, T., Harvey, P. H., and Ives, A. R. (1992). Procedures for the analysis of comparative data using phylogenetically independent contrasts. Systematic Biology 41, 18–32.
Goodwin, N. B., Dulvy, N. K., and Reynolds, J. D. (2002). Life-history correlates of the evolution of live bearing in fishes. Philosophical Transactions of the Royal Society B. Biological Sciences 357, 259–267.
| Life-history correlates of the evolution of live bearing in fishes.Crossref | GoogleScholarGoogle Scholar |
Goodwin, N. B., Dulvy, N. K., and Reynolds, J. D. (2005). Macroecology of live-bearing in fishes: latitudinal and depth range comparisons with egg-laying relatives. Oikos 110, 209–218.
| Macroecology of live-bearing in fishes: latitudinal and depth range comparisons with egg-laying relatives.Crossref | GoogleScholarGoogle Scholar |
Goto, T. (2001). Comparative anatomy, phylogeny and cladistic classification of the order Orectilobiformes (Chondrichthyes, Elasmobranchii). Memoirs of the Graduate School of Fisheries Science at Hokkaido University 48, 1–100.
Hilborn, R., and Mangel, M. (1997). ‘The Ecological Detective: Confronting Models with Data.’ (Princeton University Press: Princeton, NJ.)
Hou, C., Zuo, W. Y., Moses, M. E., Woodruff, W. H., Brown, J. H., and West, G. B. (2008). Energy uptake and allocation during ontogeny. Science 322, 736–739.
| Energy uptake and allocation during ontogeny.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BD1cXht12ltLfJ&md5=2723f6e07a48a159797f87995b1736c4CAS | 18974352PubMed |
Huber, R., vanStaaden, M. J., Kaufman, L. S., and Liem, K. F. (1997). Microhabitat use, trophic patterns, and the evolution of brain structure in African cichlids. Brain, Behavior and Evolution 50, 167–182.
| Microhabitat use, trophic patterns, and the evolution of brain structure in African cichlids.Crossref | GoogleScholarGoogle Scholar | 1:STN:280:DyaK2svjsVGgsg%3D%3D&md5=be3bc6a11f1e08f128c4f7b36ba4a4d4CAS | 9288416PubMed |
Isler, K., and van Schaik, C. P. (2009). The expensive brain: a framework for explaining evolutionary changes in brain size. Journal of Human Evolution 57, 392–400.
| The expensive brain: a framework for explaining evolutionary changes in brain size.Crossref | GoogleScholarGoogle Scholar | 19732937PubMed |
Ito, H., Yoshimoto, M., and Somiya, H. (1999). External brain form and cranial nerves of the megamouth shark, Megachasma pelagios. Copeia , 210–213.
| External brain form and cranial nerves of the megamouth shark, Megachasma pelagios.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DyaK1MXhs1yqtLk%3D&md5=443cd7abe0d7ae22611ed5feb58f0639CAS |
Ives, A. R., Midford, P. E., and Garland, T. (2007). Within-species variation and measurement error in phylogenetic comparative methods. Systematic Biology 56, 252–270.
| Within-species variation and measurement error in phylogenetic comparative methods.Crossref | GoogleScholarGoogle Scholar | 17464881PubMed |
Iwaniuk, A. N., and Nelson, J. E. (2001). A comparative analysis of relative brain size in waterfowl (Anseriformes). Brain, Behavior and Evolution 57, 87–97.
| A comparative analysis of relative brain size in waterfowl (Anseriformes).Crossref | GoogleScholarGoogle Scholar | 1:STN:280:DC%2BD3MznvVynsQ%3D%3D&md5=bdcc6c791b753bf442b85c6f5fb53a1bCAS | 11435669PubMed |
Kotrschal, K., Van Staaden, M. J., and Huber, R. (1998). Fish brains: evolution and environmental relationships. Reviews in Fish Biology and Fisheries 8, 373–408.
| Fish brains: evolution and environmental relationships.Crossref | GoogleScholarGoogle Scholar |
Kruska, D. C. T. (1988). The brain of the basking shark (Cetorhinus maximus). Brain, Behavior and Evolution 32, 353–363.
| The brain of the basking shark (Cetorhinus maximus).Crossref | GoogleScholarGoogle Scholar | 1:STN:280:DyaL1M7ltV2gsw%3D%3D&md5=ea4efb52cf7b0ce5a2293c58ac8dac83CAS | 3228691PubMed |
Last, P. R. (2007). The state of chondrichthyan taxonomy and systematics. Marine and Freshwater Research 58, 7–9.
| The state of chondrichthyan taxonomy and systematics.Crossref | GoogleScholarGoogle Scholar |
Last, P., and Stevens, J. (2009). ‘Sharks and Rays of Australia.’ (Harvard University Press: Cambridge, MA.)
Lefebvre, L., Nicolakakis, N., and Boire, D. (2002). Tools and brains in birds. Behaviour 139, 939–973.
| Tools and brains in birds.Crossref | GoogleScholarGoogle Scholar |
Lisney, T. J., Yopak, K. E., Montgomery, J. C., and Collin, S. P. (2008). Variation in brain organization and cerebellar foliation in chondrichthyans: batoids. Brain, Behavior and Evolution 72, 262–282.
| Variation in brain organization and cerebellar foliation in chondrichthyans: batoids.Crossref | GoogleScholarGoogle Scholar | 19001808PubMed |
Maddison, W., and Maddison, D. (2010). ‘Mesquite: A Modular System for Evolutionary Analysis v 2.73.’ Available at http://mesquiteproject.org [accessed September 2009].
Martin, R. D. (1981). Relative brain size and basal metabolic-rate in terrestrial vertebrates. Nature 293, 57–60.
| Relative brain size and basal metabolic-rate in terrestrial vertebrates.Crossref | GoogleScholarGoogle Scholar | 1:STN:280:DyaL3M3ntVOmtA%3D%3D&md5=1df785ff6f8f36ea26da5d8a97b91703CAS | 7266659PubMed |
Martin, R. D. (1996). Scaling of the mammalian brain: the maternal energy hypothesis. News in Physiological Sciences 11, 149–156.
Martin, A. P., Naylor, G. J. P., and Palumbi, S. R. (1992). Rates of mitochondrial DNA evolution i sharks are slow compared with mammals. Nature 357, 153–155.
| Rates of mitochondrial DNA evolution i sharks are slow compared with mammals.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DyaK38XktVCmtLg%3D&md5=a41faf1d0532ec43ce905bd70ed7c122CAS | 1579163PubMed |
McEachran, J., and Aschliman, N. (2004). Phylogeny of Batiodea. In ‘Biology of Sharks and Their Relatives’. (Eds J. Carrier, J. Musick and M. Heithaus.) pp. 79–113. (CRC Press: New York.)
Musick, J., and Ellis, J. (2005). Reproductive evolution of chondrichthyans. In ‘Reproductive Biology and Phylogeny of Chondrichthyes: Sharks, Batoids and Chimaeras’. (Eds W. Hamlett and B. Jamieson.) pp. 45–79. (Science Publishers, Inc.: Enfield, NH.)
Naylor, G. J. P. (1992). The phylogenetic relationships among requiem and hammerhead sharks: inferring phylogeny when thousands of equally most parsimonious trees result. Cladistics – the International Journal of the Willi Hennig Society 8, 295–318.
Naylor, G. J. P., Ryburn, J. A., Fedrigo, O., and Lopez, A. (2005). Phylogenetic relationships among the major lineages of modern elasmobranchs. In ‘Reproductive Biology and Phylogeny of Chondrichthyans: Sharks, Skates, Rays and Chimaeras’. (Eds W. C. Hamlett and B. Jamieson.) pp. 1–25. (Science Publishers, Inc.: Enfield, NH.)
Northcutt, R. G. (1977). Elasmobranch central nervous-system organization and its possible evolutionary significance. American Zoologist 17, 411–429.
Northcutt, R. G. (1978). Brain organization in the cartilaginous fishes. In ‘Sensory Biology of the Sharks, Skate, and Rays’. (Eds E. S. Hodgson and R. F. Mathewson.) pp. 117–194. (Office of Naval Research: Arlington, VA.)
Northcutt, R. G. (1989). Brain variation and phylogenetic trends in elasmobranch fishes. Journal of Experimental Zoology 252, 83–100.
| Brain variation and phylogenetic trends in elasmobranch fishes.Crossref | GoogleScholarGoogle Scholar |
Pagel, M. D., and Harvey, P. H. (1988). How mammals produce large-brained offspring. Evolution 42, 948–957.
| How mammals produce large-brained offspring.Crossref | GoogleScholarGoogle Scholar |
Paradis, E. (2006). ‘Analysis of Phylogenetics with R.’ (Springer: New York.)
Reynolds, J. D., Goodwin, N. B., and Freckleton, R. P. (2002). Evolutionary transitions in parental care and live bearing in vertebrates. Philosophical Transactions of the Royal Society B. Biological Sciences 357, 269–281.
| Evolutionary transitions in parental care and live bearing in vertebrates.Crossref | GoogleScholarGoogle Scholar |
Reznick, D., Meredith, R., and Collette, B. B. (2007). Independent evolution of complex life history adaptations in two families of fishes, live-bearing halfbeaks (zenarchopteridae, beloniformes) and poeciliidae (cyprinodontiformes). Evolution 61, 2570–2583.
| Independent evolution of complex life history adaptations in two families of fishes, live-bearing halfbeaks (zenarchopteridae, beloniformes) and poeciliidae (cyprinodontiformes).Crossref | GoogleScholarGoogle Scholar | 17725625PubMed |
Rosenberger, L. J. (2001). Phylogenetic relationships within the stingray genus Dasyatis (Chondrichthyes: Dasyatidae). Copeia , 615–627.
| Phylogenetic relationships within the stingray genus Dasyatis (Chondrichthyes: Dasyatidae).Crossref | GoogleScholarGoogle Scholar |
Sanderson, M. J. (2002). Estimating absolute rates of molecular evolution and divergence times: a penalized likelihood approach. Molecular Biology and Evolution 19, 101–109.
| 1:CAS:528:DC%2BD38XhtFCitQ%3D%3D&md5=1a7a7ad8754a01b005f9173f4431baf2CAS | 11752195PubMed |
Shine, R. (1989). Ecological influences on the evolution of vertebrate viviparity. In ‘Complex Organismal Functions: Integration and Evolution in Vertebrates’. (Eds D. B. Wake and G. Roth.) pp. 263–278. (Wiley-Interscience: New York.)
Shine, R. (2005). Life-history evolution in reptiles. Annual Review of Ecology Evolution and Systematics 36, 23–46.
| Life-history evolution in reptiles.Crossref | GoogleScholarGoogle Scholar |
Shirai, S. (1992). ‘Squalean Phylogeny: A New Framework of ‘Squaloid’ Sharks and Related Taxa.’ (Hokkaido University Press: Sapporo, Japan.)
Shirai, S. (1996). Phylogenetic relationships of the angel sharks, with comments of elasmobranch phyloheny (Chondrichthyes, Squatinidae). Copeia , 505–518.
Shumway, C. A. (2008). Habitat complexity, brain, and behavior. Brain, Behavior and Evolution 72, 123–134.
| Habitat complexity, brain, and behavior.Crossref | GoogleScholarGoogle Scholar | 18836258PubMed |
Smith, R. J. (2009). Use and misuse of the reduced major axis for line-fitting. American Journal of Physical Anthropology 140, 476–486.
| Use and misuse of the reduced major axis for line-fitting.Crossref | GoogleScholarGoogle Scholar | 19425097PubMed |
Smith, C. C., and Fretwell, S. D. (1974). Optimal balance between size and number of offspring. American Naturalist 108, 499–506.
| Optimal balance between size and number of offspring.Crossref | GoogleScholarGoogle Scholar |
Striedter, G. (2005). ‘Principles of Brain Evolution.’ (Sinauer Associates, Inc.: Sutherland, MA.)
Vélez-Zuazo, X., and Agnarsson, I. (2010). Shark tales: a molecular species-level phylogeny of sharks (Selachimorpha, Chondrichthyes). Molecular Phylogenetics and Evolution 52, 207–217.
Wourms, J. P. (1977). Reproduction and development in chondrichthyan fishes. American Zoologist 17, 379–410.
Wourms, J. P. (1981). Viviparity – the maternal-fetal relaionship in fishes. American Zoologist 21, 473–515.
Wourms, J. P. (1993). Maximization of evolutionary trends for placental viviparity in the spadenose shark, Scoliodon laticaudus. Environmental Biology of Fishes 38, 269–294.
| Maximization of evolutionary trends for placental viviparity in the spadenose shark, Scoliodon laticaudus.Crossref | GoogleScholarGoogle Scholar |
Wourms, J. P. (1994). The challenges of piscine viviparity. Israel Journal of Zoology 40, 551–568.
Wourms, J. P., and Demski, L. S. (1993). The reproduction and development of sharks, skates, rays, and ratfishes – introduction, history, overview, and future-prospects. Environmental Biology of Fishes 38, 7–21.
| The reproduction and development of sharks, skates, rays, and ratfishes – introduction, history, overview, and future-prospects.Crossref | GoogleScholarGoogle Scholar |
Wourms, J. P., and Lombardi, J. (1992). Reflections of the evolution of piscine viviparity. American Zoologist 32, 276–293.
Yopak, K. E., and Frank, L. R. (2009). Brain size and brain organization of the whale shark, Rhincodon typus, using magnetic resonance imaging. Brain, Behavior and Evolution 74, 121–142.
| Brain size and brain organization of the whale shark, Rhincodon typus, using magnetic resonance imaging.Crossref | GoogleScholarGoogle Scholar | 19729899PubMed |
Yopak, K. E., and Montgomery, J. C. (2008). Brain organization and specialization in deep-sea chondrichthyans. Brain, Behavior and Evolution 71, 287–304.
| Brain organization and specialization in deep-sea chondrichthyans.Crossref | GoogleScholarGoogle Scholar | 18431055PubMed |
Yopak, K. E., Lisney, T. J., Collin, S. P., and Montgomery, J. C. (2007). Variation in brain organization and cerebellar foliation in chondrichthyans: sharks and holocephalans. Brain, Behavior and Evolution 69, 280–300.
| Variation in brain organization and cerebellar foliation in chondrichthyans: sharks and holocephalans.Crossref | GoogleScholarGoogle Scholar | 17314475PubMed |
Yopak, K. E., Lisney, T. J., Darlington, R. B., Collin, S. P., Montgomery, J. C., and Finlay, B. L. (2010). A conserved pattern of brain scaling from sharks to primates. Proceedings of the National Academy of Sciences, USA 107, 12 946–12 951.
| A conserved pattern of brain scaling from sharks to primates.Crossref | GoogleScholarGoogle Scholar | 1:CAS:528:DC%2BC3cXpsVCrs7c%3D&md5=29f58b7b2b0d2e24333619b37f3a429eCAS |