Peronospora variabilis on Chenopodium murale in India
Pankaj Baiswar A C , Satish Chandra A , Rajesh Kumar B and S. V. Ngachan AA Division of Plant Pathology, ICAR Research Complex for NEH Region, Umiam-793103, Meghalaya, India.
B Division of Agronomy, ICAR Research Complex for NEH Region, Umiam-793103, Meghalaya, India.
C Corresponding author. Email: pbaiswar@yahoo.com
Australasian Plant Disease Notes 5(1) 45-47 https://doi.org/10.1071/DN10017
Submitted: 28 March 2010 Accepted: 10 May 2010 Published: 24 May 2010
Abstract
Downy mildew was observed on Chenopodium murale at Barapani, Meghalaya, India. C. murale is used as a leafy vegetable in the north-eastern hills region of India. Light and scanning electron microscopy revealed the presence of Peronospora variabilis.
Chenopodium murale, also known as nettle-leaved goosefoot belongs to the family Chenopodiaceae. This annual herb is native to parts of Asia, Africa and Europe. In north-east India tribal communities use it as a leafy vegetable. Many ethnic communities also use the leaves of Chenopodium for curing urinary problems and also for removing intestinal worms. The leaves are considered a rich and inexpensive source of protein, carotenoids and vitamins. This is considered as a potential crop since it can thrive in adverse situations with minimum agricultural input. It also has potential for use in bioremediation programs as it can accumulate large amounts of heavy metals (Bhargava et al. 2008 and references cited therein).
Typical downy mildew symptoms were observed on C. murale in 2009. Symptoms included greyish felt-like growth with corresponding yellow lesions on underside of both mature and immature leaves. Heavily infected leaves turned necrotic. Surveys in nearby areas also revealed the widespread occurrence of this disease. Voucher specimens have been deposited in the herbarium collection of MACS Agharkar Research Institute, Pune, India and ICAR Research Complex for NEH Region, India (AMH No. 9327, ICARHNEH-120).
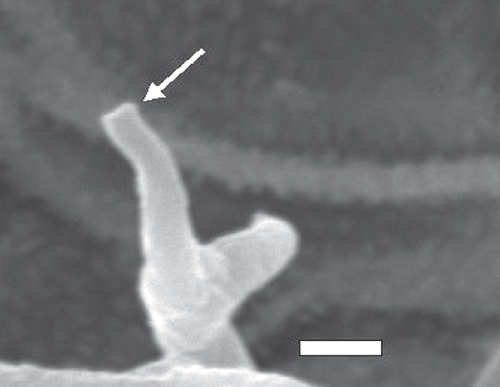
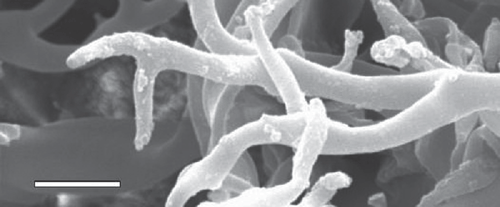
Light microscopy was done using 3% potassium hydroxide (KOH) as mounting media. Fifty measurements were made following the recommendation of Hamilton and Cunnington (2006). Areas containing greyish growth were selected using a dissecting microscope, and were cut and placed on double-sided cellotape then sputter-coated with gold using Fine Coat Ion Sputter JFC–1100 (JEOL, Tokyo, Japan). Gold-coated samples were then placed on aluminium stubs in JEOL JSM 6360 (JEOL, Tokyo, Japan). Observations were made regarding the broadening of conidiophores toward ramifications and fine structure of ultimate branchlets (Thines 2006).
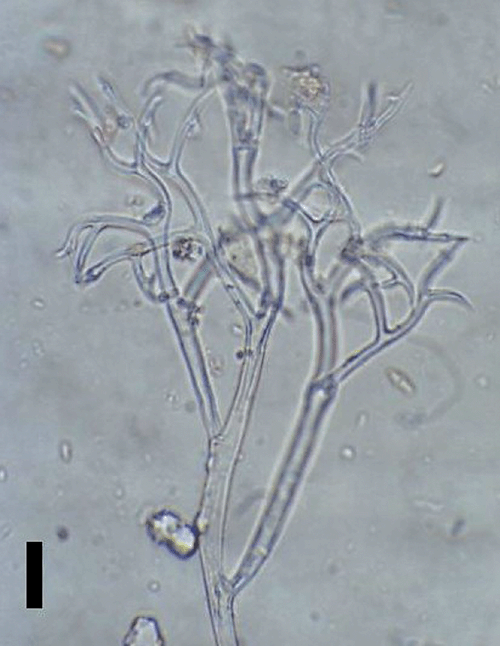
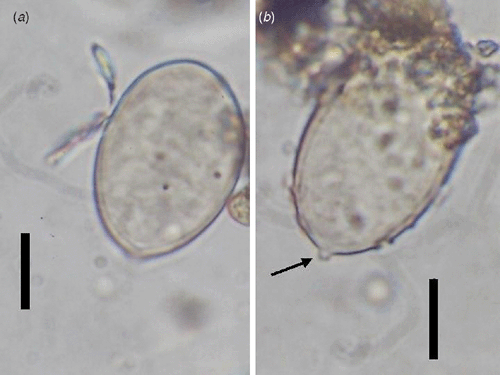
Conidiophores were straight or curved and measured 390–810 μm and trunks were 181.5–513 μm long. Callose plugs were absent. Branching was monopodial, ultimate branchlets were in pairs, curved, measuring 6.5–16 μm and widths at the base were 2–4 μm (Fig. 1). Conidia were pale brown, broadly ellipsoidal to ellipsoidal measuring 25.5–32.5 × 18–21 μm (length : breadth ratio, 1.2 : 1.7). Pedicel was also present on conidia (Fig. 2). Scanning electron microscopy observations revealed truncate conical tips of ultimate branchlets and widening of conidiophores toward ramifications (Figs 3, 4). Based on these morphological characters the pathogen was identified as Peronospora variabilis (Choi et al. 2008, 2010). Resting organs were not observed. Pathogenicity tests yielded positive results.
|
|
|
|
Previously, a broad species concept for the Peronospora species parasitic on Chenopodiaceae prevailed, with all considered identical to P. farinosa (Yerkes and Shaw 1959). More recently, a narrow species concept has been proven in case of Peronospora infecting Chenopodiaceae (Choi et al. 2007, 2008). They suggested that P. variabilis, P. boni-henrici, P. chenopodii, and P. chenopodii-polyspermi are four downy mildew pathogens specific to C. album, C. bonus-henricus, C. hybridum and C. polyspermum, respectively. P. muralis has also been reported on C. murale but conidia of P. muralis are subglobose to broadly ellipsoidal and without pedicels (Gäumann 1923).
Following the broad species concept, P. farinosa f. sp. chenopodii and P. farinosa have been reported on C. quinoa and C. murale, respectively from India (Verma et al. 1964; Kumar et al. 2006). Since both these reports were based on a broad species concept, they did not mention any morphological information related to presence or absence of pedicels and range of length : breadth ratio of the conidia, which have emerged to be important characters for delimitation of Peronospora species parasitic on Chenopodium spp. (Choi et al. 2008, 2010). Possibility of two different species infecting C. murale cannot be denied since the conidial dimensions provided by Verma et al. (1964) were very close to P. muralis.
Our report on the presence of the downy mildew pathogen P. variabilis on C. murale is the first fully described record from India. Since the correct identity of a pathogen is of utmost importance in any kind of research related to management of a disease, we hope this report will be of help in the future.
Acknowledgements
Authors would like to thank Head of SAIF, Dr Sudeep Dey (Scientific Officer), Dr R. Charkraborty, N. K. Rynjah for scanning electron microscopy at North-eastern Hill University, Shillong, Meghalaya, India. Authors also wish to thank the anonymous reviewer for their suggestions.
Bhargava A,
Shukla S,
Srivastava J,
Singh N, Ohri D
(2008) Genetic diversity for mineral accumulation in the foliage of Chenopodium spp. Scientia Horticulturae 118, 338–346.
| Crossref | GoogleScholarGoogle Scholar |
CAS |

Choi YJ,
Hong SB, Shin HD
(2007) Re-consideration of Peronospora farinosa infecting Spinacia oleracea as distinct species, Peronospora effusa. Mycological Research 111, 381–391.
| Crossref | GoogleScholarGoogle Scholar |
CAS |
PubMed |

Choi YJ,
Denchev CM, Shin HD
(2008) Morphological and molecular analyses support the existence of host-specific Peronospora species infecting Chenopodium. Mycopathologia 165, 155–164.
| Crossref | GoogleScholarGoogle Scholar |
CAS |
PubMed |

Choi YJ,
Danielsen S,
Lübeck M,
Hong SB,
Delhey R, Shin HD
(2010) Morphological and molecular characterization of the causal agent of downy mildew on quinoa (Chenopodium quinoa). Mycopathologia 169, 403–412.
| Crossref | GoogleScholarGoogle Scholar |
CAS |
PubMed |

Gäumann E
(1923) Beiträge zu einer Monographie der Gattung Peronospora Corda. Beitrage Krypto-gamen. Flora der Schweiz 5, 1–360.

Hamilton AJ, Cunnington JH
(2006) Calculating minimum sample sizes for taxonomic measurements: examples using Gäumann’s Peronospora spore data. Mycotaxon 95, 189–194.

Kumar A,
Bharagava A,
Shukla S,
Singh HB, Ohri D
(2006) Screening of exotic Chenopodium quinoa accessions for downy mildew resistance under mid-eastern conditions of India. Crop Protection (Guildford, Surrey) 25, 879–889.
| Crossref | GoogleScholarGoogle Scholar |

Thines M
(2006) Evaluation of characters available from herbarium vouchers for the phylogeny of the downy mildew genera (Chromista, Peronosporales), with focus on scanning electron microscopy. Mycotaxon 97, 195–218.

Verma SC,
Chauhan LS, Mathur RS
(1964) Peronospora farinose (Fr.) Fr. on Chenopodium murale L. – A new record for India. Current Science 33, 720–721.

Yerkes WD, Shaw CG
(1959) Taxonomy of the Peronospora species on Cruciferae and Chenopodiaceae. Phytopathology 49, 499–507.



